Detergent-free Membrane Protein Expression
Detergent, one kind of surfactant, can disrupt the phospholipid bilayer of the membrane into small micelles and induce protein inactivation during conventional membrane protein production. Therefore, detergent-free production of membrane protein with their native conformation retained has significant advantages in structural and functional studies. Creative Biostructure endeavors to develop water-soluble membrane protein production in the absence of detergent. We employ several techniques to perform Mempro™ detergent-free membrane protein production.
- Nanodisc
Nanodiscs are a synthetic model membrane system that is composed of lipid bilayer of phospholipids with the hydrophobic side screened by two membrane scaffolding proteins (MSPs) aligned in double belt formation. Membrane proteins can be stabilized in nanodiscs without the need for any detergents (Figure 1). This technology is widely applied for solubilization and characterization of integral membrane proteins, such as G-protein coupled receptors (GPCRs).
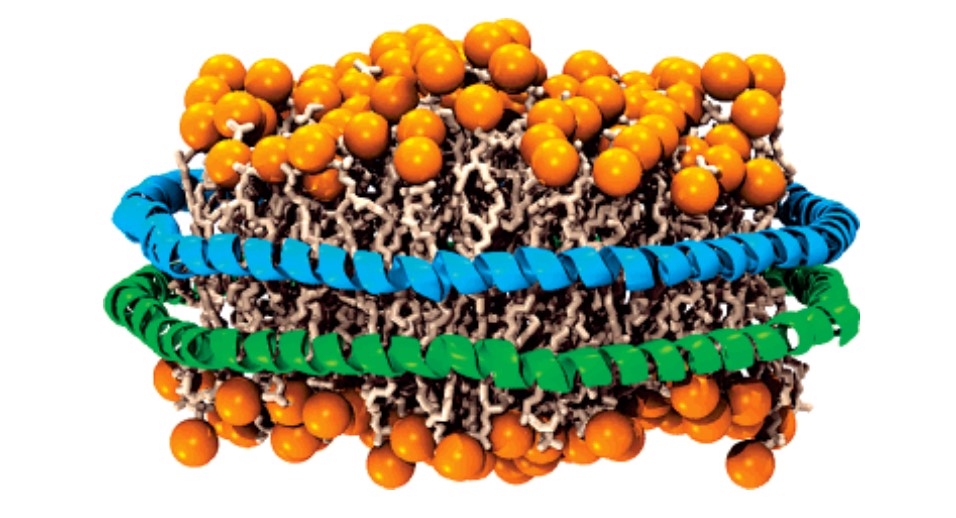 Figure 1. Schematic diagram of a nanodisc.
Figure 1. Schematic diagram of a nanodisc.
- Amphipol
Amphipols, including A8-35, A8-75, A34-75 and A34-45, can replace the detergent used to keep membrane proteins soluble in aqueous solutions. The amphipathic amphipols have the ability to enwrap the transmembrane regions of proteins, allowing them to stay folded (Figure 2). The amphipols can be applied in electron microscopy (EM), nuclear magnetic resonance (NMR), in vitro functional analysis and other potential utilizations involving membrane proteins.
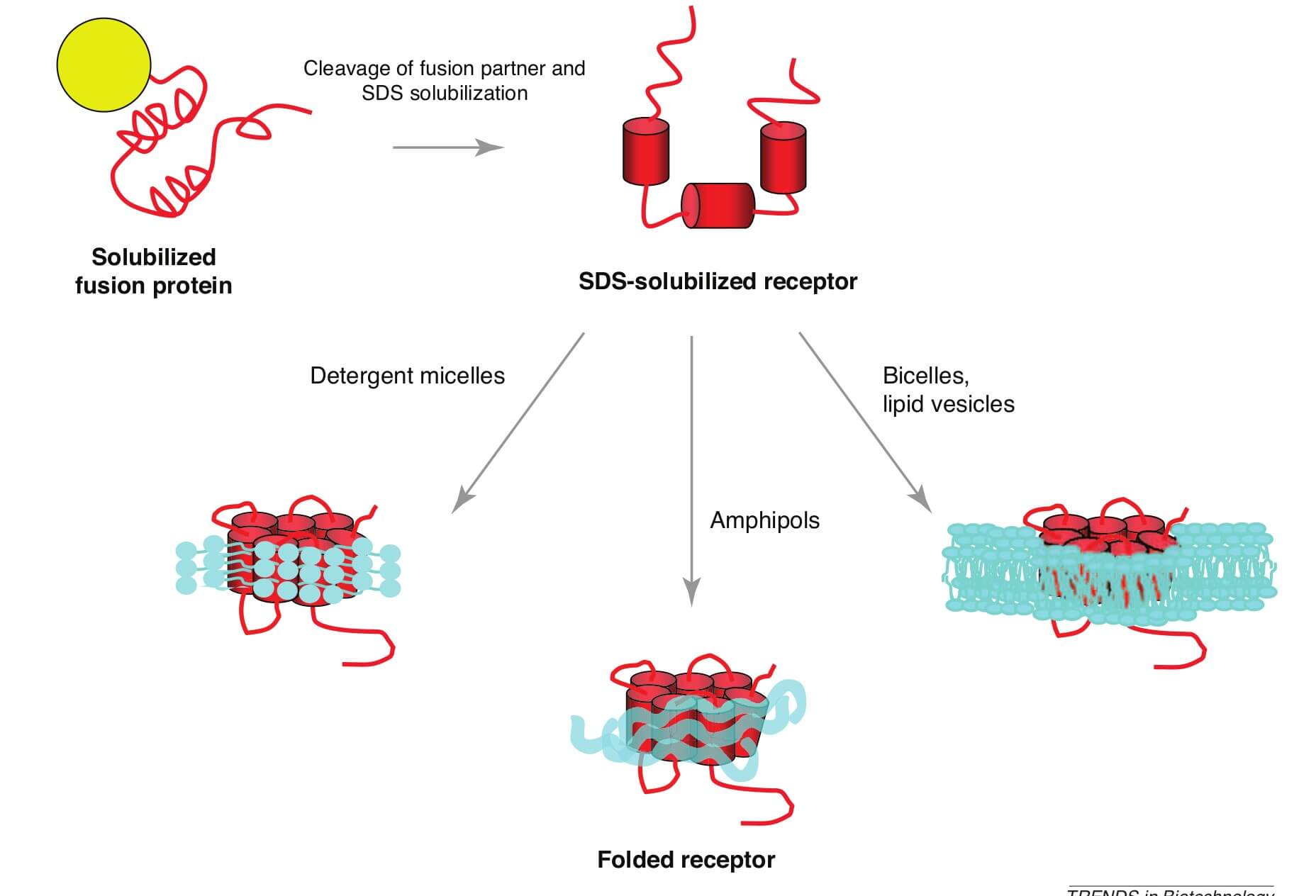 Figure 2. Comparison between amphipols and detergents.
Figure 2. Comparison between amphipols and detergents.
- SMALP
Poly(styrene-co-maleic acid) lipid particles (SMALPs) can reversibly encapsulate membrane proteins (Figure 3). The polymer itself is made of alternating hydrophilic (maleic acid) and hydrophobic (styrene) moieties. The SMALPs are self-assembled simply by the addition of the SMA co-polymer. SMALP is an ideal system suitable for membrane protein purification and further biochemical studies (such as drug discovery) due to its high temperature resistance and pH sensitivity.
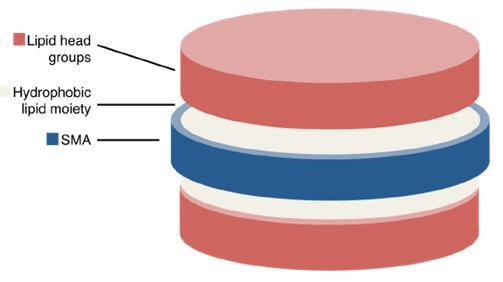 Figure 3. Diagrammatic representation of a disc-like SMALP.
Figure 3. Diagrammatic representation of a disc-like SMALP.
Creative Biostructure provides unparalleled membrane protein production services using Mempro™ detergent-free methodologies, which enable comprehensively structural and functional studies on membrane proteins of your interest. Please see the list of membrane proteins provided by Creative Biostructure.
Please feel free to contact us for a detailed quote!
Ordering Process
References
- Baneres JL, et al. (2011) “New advances in production and functional folding of G-protein-coupled receptors”. Trends Biotechnol 29(7):314-322.
- Jamshad M, et al. (2011) “Surfactant-free purification of membrane proteins with intact native membrane environment”. Biochem Soc Trans 39:813-818.
- Ryan RO (2010) “Nanobiotechnology applications of reconstituted high density lipoprotein”. J Nanobiotechnol 8(1):28-37.
- Shih AY, et al. (2007) “Disassembly of nanodiscs with cholate”. Nano Lett 7(6): 1692-1696.
- Tribet C, et al. (1996) “Amphipols: polymers that keep membrane proteins soluble in aqueous solutions”. Proc Natl Acad Sci U. S. A. 93:15047-15050.
