Fluorescent Exosomes/Microvesicles
The fluorescent exosomes/microvesicles are now an efficient new tool for intercellular communication research, diagnostics and drug design. Creative Biostructure offers fluorescently labeled exosomes from various species, tissues and cell lines from healthy and diseased donors. Such exosomes still have the specialized properties of their parent cells, and are readily available in standardized formats for flow cytometry (FACS), western blotting (WB) and enzyme-linked immunosorbent assay (ELISA). Our products are control standards for biological functions and support academic and industrial research. Apart from these products, we also offer custom exosome isolation services and labeling services for exosomes in accordance with the experimental requirements.
Product List
Background
These are the two main kinds of EVs—exosomes and microvesicles—that mediate cell-cell interaction involved in many physiological and pathological processes. The endosomal pathway produces exosomes which are released when MVBs connect with the cell membrane. Microvesicles (also known as ectosomes or microparticles), however, are released directly from the cell membrane. Both EVs carry bioactive molecules—proteins, nucleic acids, lipids and metabolites—indicative of the health and function of their parent cells. This unique cargo makes them especially valuable for studying how cells function and how diseases progress.
Advanced techniques of visualization and tracking of exosomes and microvesicles include methods such as fluorescent dyes, gene tags, antibody tagging. For example, labeling exosomes through lentivirus-mediated CD63-GFP expression and genetic tagging with fluorescent proteins. Subsequently, the fluorescent-labeled vesicles may be studied by flow cytometry, confocal microscopy, and in vivo imaging. These techniques are enabling the modeling of vesicle behavior both in cellular dynamics and disease progression.
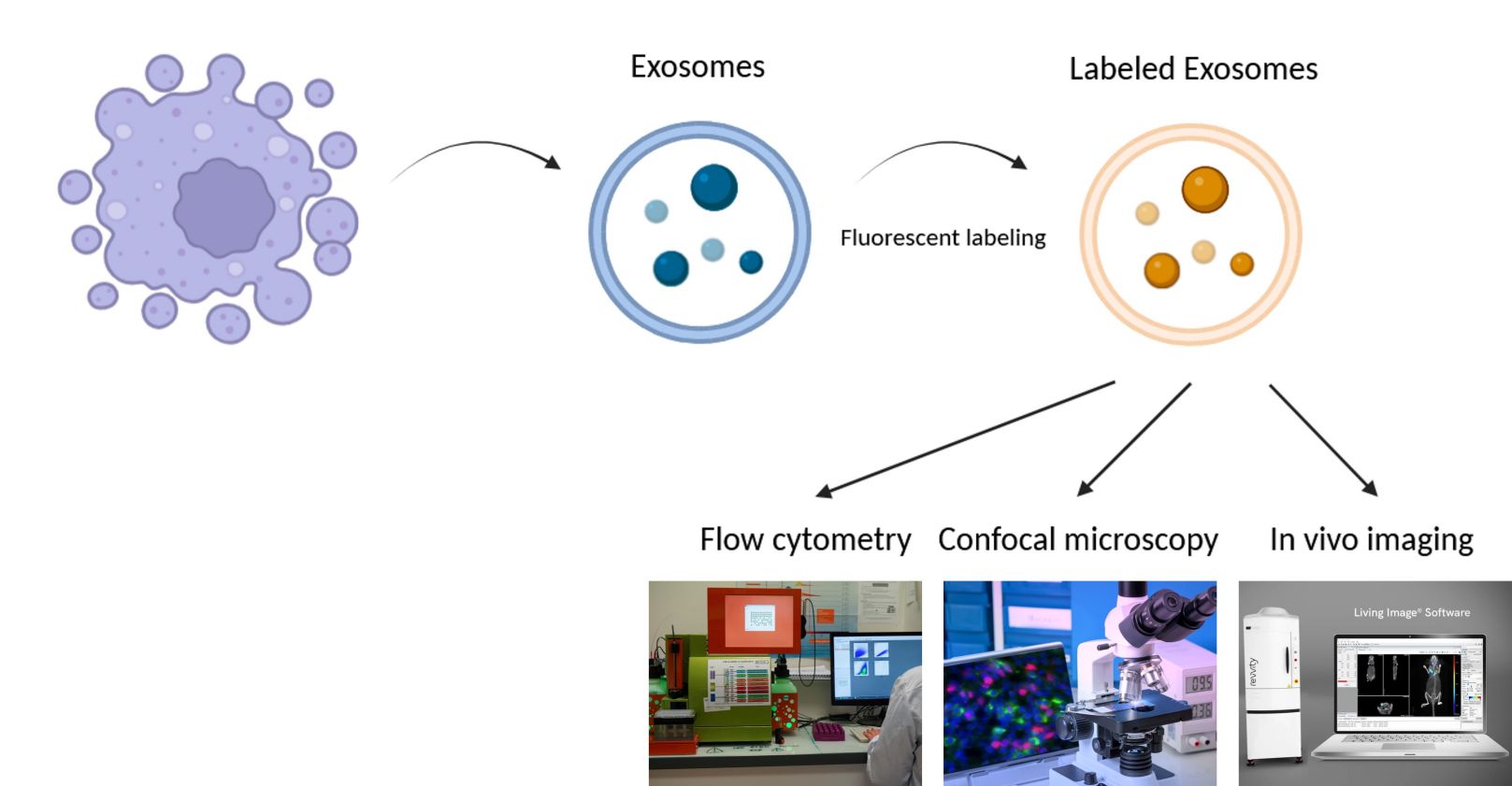 Figure 1. Visualize and track vesicles with fluorescent labeling.
Figure 1. Visualize and track vesicles with fluorescent labeling.
Applications of Fluorescent Exosomes/Microvesicles
Diagnostics and Biomarker Discovery
Diagnostics and biomarker detection are two of the promising uses for fluorescently tagged exosomes and microvesicles. Because they are stable in bodily fluids and borne of molecular signatures, such vesicles might serve as cancer markers, heart disease markers, and neurodegenerative markers. Disease-related molecular markers can be detected from patient exosomes labeled with fluorescent tags. Methods such as flow cytometry can quantitatively detect surface features and contents, leading to early detection and prognostic diagnostic biomarkers.
Drug Delivery
As drug delivery vehicles, exosomes and microvesicles are biocompatible, immune evasive and have a natural tropism for certain tissues. Fluorescent labeling can be used to monitor the biodistribution of exosome-based drug delivery systems in vivo, providing critical information on delivery rates and cellular targeting. Once filled with therapeutic cargo—small RNAs, proteins, chemotherapeutics—these tagged vesicles can be tested for effective transport to cells.
Therapeutic Target Discovery
Fluorescence-labeled exosomes and microvesicles can be used to study the onset and development of diseases and their resistance to treatment, particularly cancer. They allow researchers to follow the signaling pathways of exosomes and see how exosomes and their targets interact. Using exosomes from cancer cells, we may soon learn more about how exosomes drive tumor growth, proliferation and immune evasion. Such studies are central to identifying therapeutic targets that disrupt exosome-driven pathogenesis.
Immunology and Vaccine Development
Exosomes and microvesicles help modulate immune activity, so they're good candidates for vaccines and immunotherapy. Fluorescent labeling makes it possible to map the contact of exosomes with immune cells and determine whether this triggers immune responses. For example, antigenic peptides can be injected into exosomes isolated from immune-related cell lines and tested for immunogenicity and therapeutic efficacy in living animals. This transforms them into exosome-based vaccines targeting immune systems, opening a whole new field of vaccine development.
Cellular and Molecular Biology Research
The inherent ability of exosomes and microvesicles to communicate between cells is the ideal model for molecular interactions in networks. Using fluorescently tagged vesicles, researchers can track vesicle migration, uptake and digestion in target cells, and even perform microscope-like analyses of intercellular signaling and molecular trafficking. This has enabled us to better model cellular inflammation, repair and neuroprotection.
Case Studies
Case Study 1: Covalently Labeled Fluorescent Exosomes for In Vitro and In Vivo Applications
This study presents a novel method for covalent attachment of commercial fluorescent dyes to exosome surfaces, which was tested on milk and cancer cell-derived exosomes (U87 and B16F10). Fluorescent labeling was found to preserve the original physicochemical properties of the exosomes. Validation in cell culture and healthy mice showed successful visualization of labeled exosomes by optical imaging, revealing their biodistribution over time without animal sacrifice. Milk exosomes accumulated primarily in the liver and spleen, confirming their retention by Kupffer macrophages. Importantly, histologic evaluation revealed no tissue damage. The methodology was robust to different dyes and exosome types, and confocal imaging confirmed internalization in hepatocytes. These results suggest that fluorescently labeled natural exosomes may serve as effective alternatives to synthetic nanoparticles for non-invasive therapeutic applications.
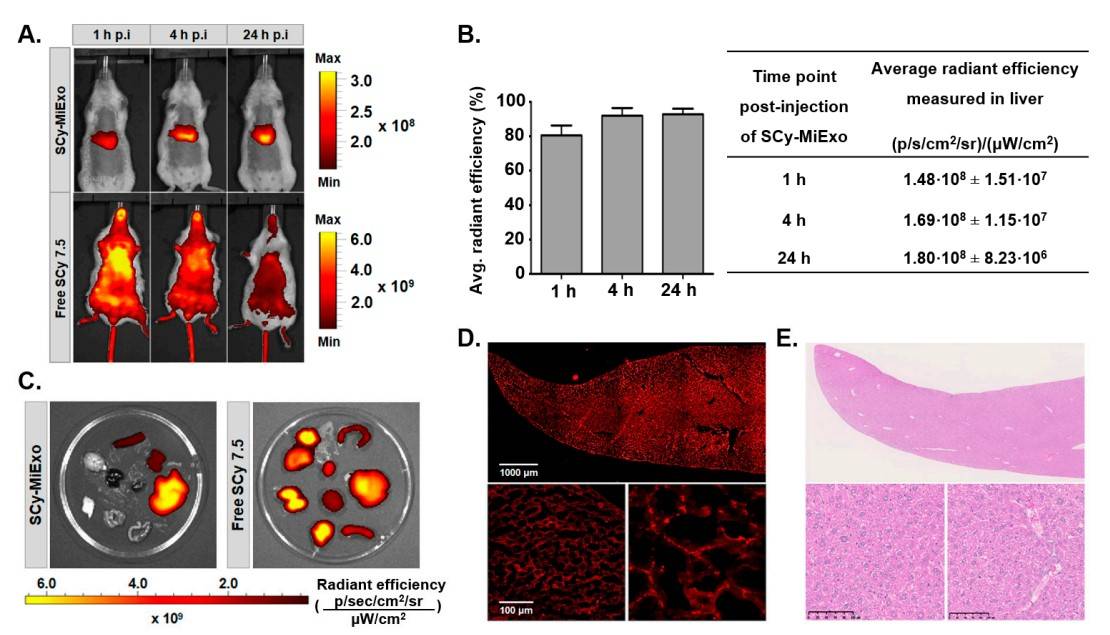 Figure 2. In vivo and ex vivo studies with sulfo-cyanine 7.5-labeled milk exosomes (SCy-MiExo): (A) In vivo optical imaging of sulfo-cyanine 7.5-labeled milk exosomes (SCy-MiExo, top) and free SCy 7.5 (bottom) in healthy mice. (B) The time course of average radiant efficiency measured in vivo in livers of mice treated with SCy-MiExo. The data are expressed as the % (graph) and in units of p/s/cm2/sr)/(µW/cm2), with the mean ± standard deviation. (C) Ex vivo biodistribution of SCy-MiExo (left) and free SCy 7.5 (right) in excised organs. (D) Confocal images of liver sections from mice treated with SCy-MiExo. Right down image presents a zoom of left down image. (E) Hematoxylin and Eosin (H&E) histological images of liver sections from mice treated with SCy-MiExo. (González MI, et al., 2021)
Figure 2. In vivo and ex vivo studies with sulfo-cyanine 7.5-labeled milk exosomes (SCy-MiExo): (A) In vivo optical imaging of sulfo-cyanine 7.5-labeled milk exosomes (SCy-MiExo, top) and free SCy 7.5 (bottom) in healthy mice. (B) The time course of average radiant efficiency measured in vivo in livers of mice treated with SCy-MiExo. The data are expressed as the % (graph) and in units of p/s/cm2/sr)/(µW/cm2), with the mean ± standard deviation. (C) Ex vivo biodistribution of SCy-MiExo (left) and free SCy 7.5 (right) in excised organs. (D) Confocal images of liver sections from mice treated with SCy-MiExo. Right down image presents a zoom of left down image. (E) Hematoxylin and Eosin (H&E) histological images of liver sections from mice treated with SCy-MiExo. (González MI, et al., 2021)
Case Study 2: Exosome-Mediated Communication in Breast Cancer Metastasis
CD4+ T-cell stimulated tumor-associated macrophages (TAMs) stimulated by IL-4 promote breast cancer invasion and metastasis. Yang group explores how exosomes secreted by TAMs could be used to infect cancer cells with microRNAs (miRNAs). In a co-culture environment, they showed that fluorescently labeled exogenous miRNAs are transported without direct contact from IL-4-activated macrophages to breast cancer cells. More strikingly, macrophage miR-223 was also increased in the SKBR3 and MDA-MB-231 cell co-cultures. By blocking miR-223 in macrophages with antisense oligonucleotides, the breast cancer cells became less invasive. Functional experiments also showed that miR-223 induces invasion through the Mef2c-β-catenin pathway. This explains why communication between exosomes is crucial for breast cancer metastasis and tells us something about the relationship between macrophages and tumor cells.
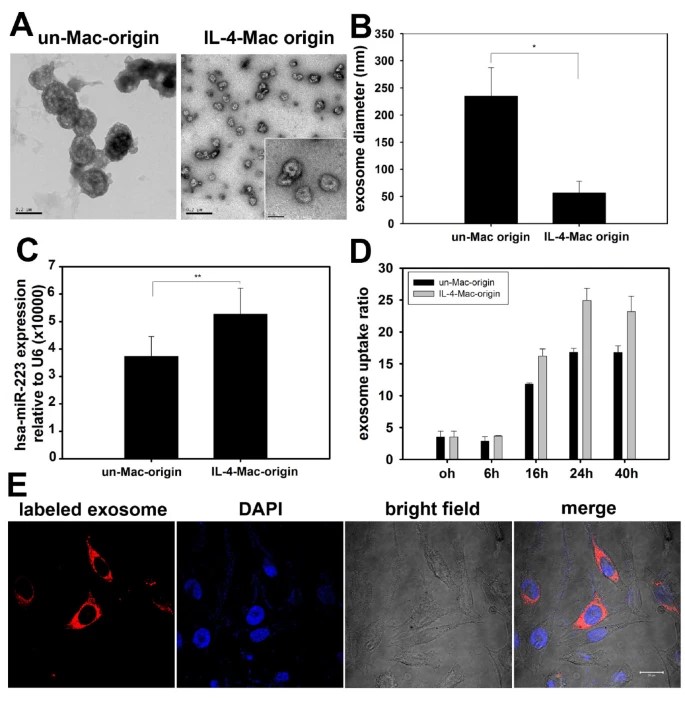 Figure 3. Exosomes secreted from macrophages shuttle miR-223 to breast cancer cells. (A) Exosomes secreted from unactivated and IL-4-activated macrophages were collected and observed with an electron microscope. Exosomes derived from unactivated macrophages (un-Mac-origin) were larger in size (100-250 nm) than those derived from IL-4-activated macrophages (IL-4-Mac-origin) (50-80 nm). (B) The diameter of exosomes derived from unactivated and IL-4-activated macrophages were quantified. Data are averages of more than 10 exosomes from each group. (C) Levels of miR-223 expression in exosomes were quantified by qRT-PCR. ** p < 0.01. (D) and (E) Exosomes derived from IL-4-activated macrophages were more readily internalized by breast cancer cells. Exosomes derived from unactivated and IL-4-activated macrophages were labeled with CM-Dil and incubated with SKBR3 cells for the indicated times (0-40 h). Uptake of exosomes by cancer cells was visualized by confocal microscopy (representative images are shown in (E) and quantified by flow cytometry in (D)). (Un-Mac, unactivated macrophages; IL4-Mac, IL-4-activated macrophages). (Yang M, et al., 2011)
Figure 3. Exosomes secreted from macrophages shuttle miR-223 to breast cancer cells. (A) Exosomes secreted from unactivated and IL-4-activated macrophages were collected and observed with an electron microscope. Exosomes derived from unactivated macrophages (un-Mac-origin) were larger in size (100-250 nm) than those derived from IL-4-activated macrophages (IL-4-Mac-origin) (50-80 nm). (B) The diameter of exosomes derived from unactivated and IL-4-activated macrophages were quantified. Data are averages of more than 10 exosomes from each group. (C) Levels of miR-223 expression in exosomes were quantified by qRT-PCR. ** p < 0.01. (D) and (E) Exosomes derived from IL-4-activated macrophages were more readily internalized by breast cancer cells. Exosomes derived from unactivated and IL-4-activated macrophages were labeled with CM-Dil and incubated with SKBR3 cells for the indicated times (0-40 h). Uptake of exosomes by cancer cells was visualized by confocal microscopy (representative images are shown in (E) and quantified by flow cytometry in (D)). (Un-Mac, unactivated macrophages; IL4-Mac, IL-4-activated macrophages). (Yang M, et al., 2011)
Product Advantages
- High-Quality, Standardized Products: We offer consistently high-quality exosomes and microvesicles with reliable, standardized fluorescent labeling. Reproducible and trustworthy products are essential for accurate results in diagnostics, cellular tracking, and therapeutic studies.
- Wide Selection of Ready-to-Use Exosome Sources: We offer a comprehensive selection of ready-to-use fluorescently labeled exosome sources, ranging from healthy donors to cancerous lesions from various human and mouse cell lines, tissues, and organs. This wide array allows researchers to select standard exosomes based on their study requirements, whether for disease modeling, drug-response studies, or biomarker discovery.
- Innovation and Adaptability: Our innovation and flexibility keep us at the forefront of exosome technology. We continue to lead the way in fluorescent labeling, isolation and stability of exosomes, adapting our products to meet the new demands of cellular and molecular studies.
Resources
Frequently Asked Questions
-
Which fluorescent exosomes do you offer?
Creative Biostructure offers highly purified fluorescent exosome standards for tracking EVs during fluorescence microscopy studies. The fluorescent exosomes are labeled with a green dye for long term fluorescence labeling. They can be used for any purpose and produce a long lasting and highly visible signal.
-
What are the applications of your fluorescent exomes/microvesicles?
Our HQExo™ exosomes/microvesicles serve as reliable positive controls across a range of experimental applications. Specifically, they are highly effective in validating and calibrating assays in ELISA, FACS, and WB techniques.
-
How purified and controlled are they?
We isolate and purify exosomes using state-of-the-art isolation and purification protocols such as ultracentrifugation, size-exclusion chromatography, and immunoaffinity capture to make them as pure as possible, and all batches are rigorously quality controlled for consistency.
-
Do you offer custom exosome isolation services?
We offer exosome isolation services tailored to specific research requirements. Separations can be tailored by size, source, or other parameters for individual projects, ensuring you receive the best product for your research.
Develop your research with our ready-to-use and customizable fluorescent exosomes and microbubbles. Contact us today to find out the optimal solution!
References
- Camussi G, Deregibus MC, Bruno S, Cantaluppi V, Biancone L. Exosomes/microvesicles as a mechanism of cell-to-cell communication. Kidney International. 2010;78(9):838-848.
- González MI, González-Arjona M, Santos-Coquillat A, et al. Covalently labeled fluorescent exosomes for in vitro and in vivo applications. Biomedicines. 2021;9(1):81.
- Yang M, Chen J, Su F, et al. Microvesicles secreted by macrophages shuttle invasion-potentiating microRNAs into breast cancer cells. Mol Cancer. 2011;10(1):117.



