Multivesicular Liposomes Production
Creative Biostructure has developed a novel technology platform to offer sustained-release drug delivery – Multivesicular Liposomes. Our extensive experience in liposome manufacture can support the cutting-edge research of clinical pharmaceutics. We have demonstrated high reliability of our services after many years in serving companies and research institutes.
How Multivesicular Liposomes Work
The unique feature of the multivesicular system, a microscopic spheroid with discontinuous internal aqueous chambers bounded by a continuous, non-concentric network of lipid membrane, render a higher aqueous volume-to-lipid ratio and much larger particle diameters compared to unilamellar or multilamellar vesicular systems. The architecture of multivesicular liposomes allow encapsulating drugs with greater efficiency, provide improved structural stability, and ensure a better controlled and prolonged release of drugs. There are many favorable characteristics of exploiting multivesicular liposomes technology:
- Firstly, multivesicular vesicles compose synthetic analogues of natural lipids which makes membrane bilayers biocompatible and bio-degradable;
- Robust structural stability due to its non-concentric architecture and neutral lipids composition,
- It can achieve high drug loading with high drug recovery based on its large diameters (1-100µm) and trapped volume;
- Multivesicular liposomes provides a controlled, sustained release drug delivery over hours to weeks by manipulation of lipid and aqueous composition.
Multivesicular Vesicles Applications
Our multivesicular vesicles drug delivery technology can be a remarkable solution to target medications that conventionally require frequent injections, different administrations by the intrathecal, subcutaneous, intramuscular, intraperitoneal and intraocular routes. Our team demonstrated the versatility of this technology by encapsulating a wide range of therapeutic agents including small molecules as well as macromolecules like proteins, peptides, DNA, and oligonucleotides.
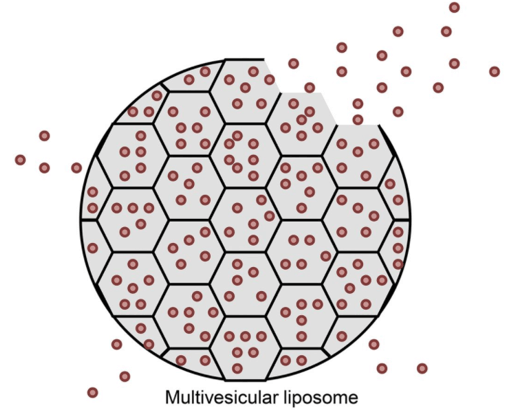 Figure 1. Morphology of multivesicular liposomes. (Pharmacological Reviews, 2016)
Figure 1. Morphology of multivesicular liposomes. (Pharmacological Reviews, 2016)
Creative Biostructure’s custom services are built on the foundation of remarkable expertise and powered by advanced equipment. We provide excellent services for preparation and production of multivesicular liposomes.
Advantages:
- High yields of encapsulation
- Accommodating high drug loading
- Various vesicle sizes
Reliable Analysis:
- Size measurement
- Encapsulation efficiency determination
- Drug integrity by HPLC testing
- In vitro drug release study
- In vivo pharmacokinetic studies
Creative Biostructure is engaged in providing the best service for liposome research. The advanced technology platform, specialized experimental design, and precise analysis make us a world-class service supplier. For more details, please contact us through online inquiry.
Ordering Process
References
- Yan Shen, Yuanxin Ji, Shengjie Xu, et al. (2011) Multivesicular liposome formulations for the sustained delivery of ropivacaine hydrochloride: Preparation, characterization, and pharmacokinetics. Drug Delivery. 18: 361–366.
- Jian Qiu, Xiao-hui Wei, Fang Geng, et al. (2005) Multivesicular liposome formulations for the sustained delivery of interferon α-2b. Acta Pharmacologica Sinica. 26: 1395–1401.
