Exosome Engineering Services
Exosomes are small lipid bilayer vesicles produced by most cells, which contain abundant nucleic acids, proteins and lipids. Exosomes have been widely used in the research of tumor therapy drugs because of their good biocompatibility, circulation ability in vivo and material delivery capacity. Invasion, metastasis and drug resistance are the main causes of tumor treatment failure. Exosomes, as the main means of intercellular transport of nucleic acids and proteins, play a key role in promoting drug resistance, invasion and metastasis of tumor cells. In addition to being the target of liquid biopsy for cancer diagnosis, exosomes also play an increasingly important role in cancer treatment, including regulation of exosome expression, use of exosomes as drug delivery system, and engineering modification of exosomes for targeted therapy.
- Exosome Cargo Loading Service
Conventional chemotherapeutic drugs (paclitaxel, tamoxifen, eccorine, etc.) have strong cytotoxicity and weak tissue selection ability, and often cause serious toxic and side effects in the treatment of cancer. Cell-derived exosomes not only have good biological compatibility but also have strong biological delivery capacity. Therefore, exosomes are gradually becoming carriers for cancer chemotherapy drug delivery. At present, the main ways of exosome drug loading include mixing, transfection reagent, ultrasonic, freeze-thaw cycle, electroporation, etc.
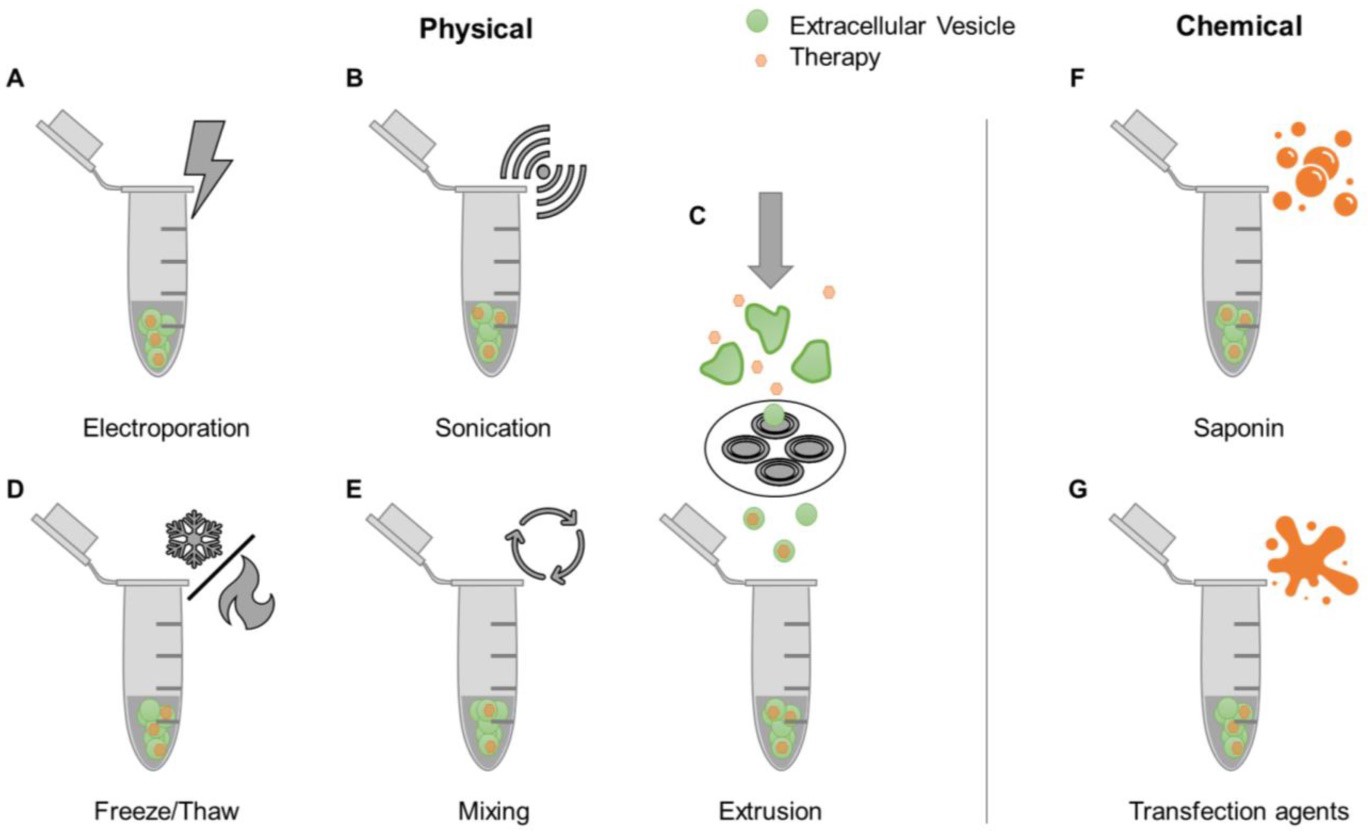 Figure 1.
Examples of drug loading methods post-EV isolation. (Walker S, et al. 2019)
Figure 1.
Examples of drug loading methods post-EV isolation. (Walker S, et al. 2019)
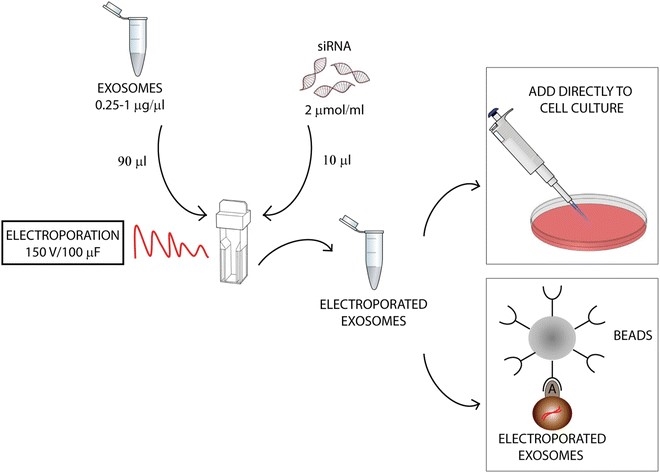 Figure 2. Electroporation of exosomes.
(Jessica Wahlgren, et al. 2016)
Figure 2. Electroporation of exosomes.
(Jessica Wahlgren, et al. 2016)
Therefore, extracellular vesicles (EVs), including exosomes, may represent a promising drug delivery strategy for exogenous small molecule and RNA-based therapies.
- Exosome Labeling Service
Exosomes contain specifical cargos, such as RNAs, lipids, and proteins. The cargos amount and composition of exosomes depend on the cell type from which they are released, which makes them useful for biomarker discovery and functional characterization. Creative Biostructure can provide multiple fluorescence labeling strategies (labeling with lipid or protein) for in vivo and in vitro visualization studies.
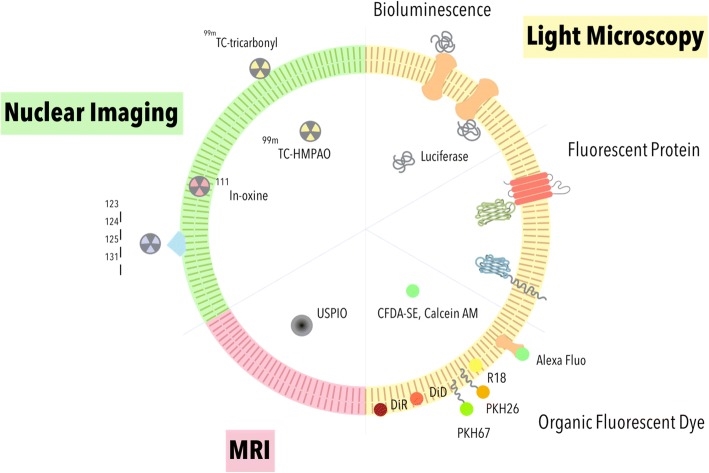 Figure 3. Strategies for
exosome labeling and imaging. (Steven Ting-Yu Chuo, et al. 2018)
Figure 3. Strategies for
exosome labeling and imaging. (Steven Ting-Yu Chuo, et al. 2018)
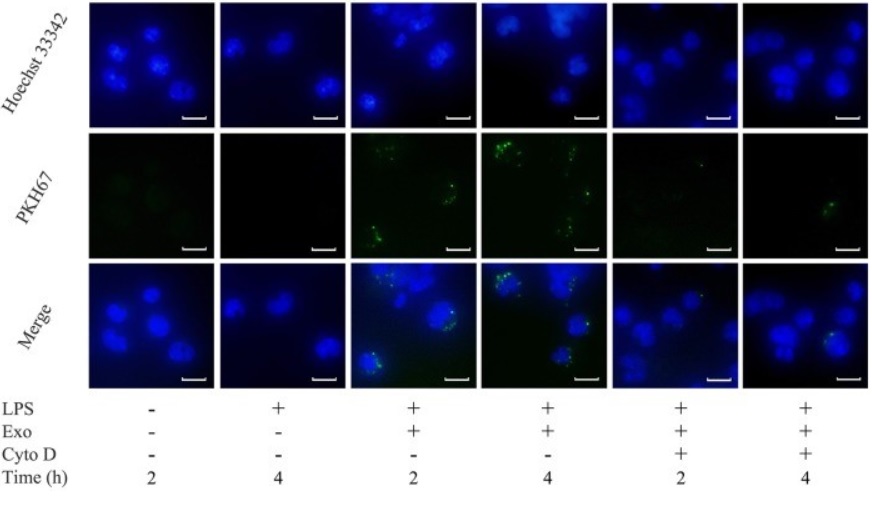 Figure 4. The uptake of exosomes by
THP-1 macrophages. (Zhuyu Wang, et al. 2019)
Figure 4. The uptake of exosomes by
THP-1 macrophages. (Zhuyu Wang, et al. 2019)
Exosome labeled with Lipophilic dyes (PKH67, green)
THP-1 macrophages labeled with nuclear staining reagent (Hoechst 33342, blue)
- Exosome Surface Display Service
Extracellular vesicles (EVs) are small membranous particles that play a crucial role in intercellular signaling and communication. Based on its molecular transfer function, high biocompatibility, and low cytotoxicity to normal tissue, exosomes become a promising carrier for therapeutic molecular delivery system. It had been reported that many membrane proteins are displayed on exosome membranes, such as CD81, CD82, CD37, CD63 and CD9, which can serve as anchoring scaffolds to directly display peptides/antigens on exosomes.
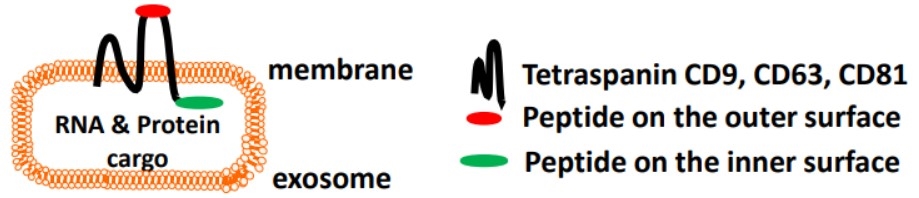 Figure 5. System
design of surface engineering of exosome. (Zachary Stickney, et al. 2016)
Figure 5. System
design of surface engineering of exosome. (Zachary Stickney, et al. 2016)
- Aptamer-Based Exosome Targeting Service
- Walker, S, et al. Extracellular vesicle-based drug delivery systems for cancer treatment. Theranostics. 2019, 9(26): 8001-8017.
- Jessica Wahlgren, et al. Delivery of small interfering RNAs to cells via exosomes. Methods Mol Biol. 2016, 1364: 105-125.
- Steven Ting-Yu Chuo, et al. Imaging extracellular vesicles: current and emerging methods. J Biomed Sci. 2018, 25: 91.
- Zhuyu Wang, et al. Cyclic stretch force induces periodontal ligament cells to secrete exosomes that suppress IL-1β production through the inhibition of the NF-κB signaling pathway in macrophages. Front Immunol. 2019, 10: 1310.
- Zachary Stickney, et al. Development of exosome surface display technology in living human cells. Biochem Biophys Res Commun. 2016, 472(1): 53-59.
Aptamer-based exosomes, a distinct subtype, are enveloped with aptamers capable of binding to specific receptors and conveying precise signals or substances. Leveraging expertise in exosome research, Creative Biostructure offers specialized services for aptamer-based exosome targeting. Our goal is to enhance the targeted delivery efficiency of exosomes through the strategic use of aptamers.
With our exosome platform, Creative Biostructure can provide the custom service for production exosome with loading therapy (proteins, nucleic acids, and chemical compounds). We focus on exosome research and are committed to providing exosome one-stop service, including exosome isolation, identification, tracking, exosome cell testing, related functional experiments such as drug carrier design and other integrated services.
If you are interested in our Exosome Engineering Services, please feel free to contact us. We are looking forward to cooperating with you.
Ordering Process
References

