Molecular Characterization
Creative Biostructure utilizes NMR spectroscopy, combined with other analytic approaches such as mass spectrometry and Fourier transformation infrared spectroscopy to characterize molecular properties of natural organic matter, polymers, unknowns, and contaminant residues.
Using simple 1H mono-dimensional spectroscopy, aliphatic, carbohydrate, aromatic, and carboxyl-rich alicyclic molecules (CRAM) can be identified by chemical shift analysis. Furthermore, 13C investigation by multi-dimensional heteronuclear approaches, for example, heteronuclear multiple quantum coherence (HMQC) and heteronuclear multiple bonding coherence (HMBC), allow characterization of specific regions assignable to well-known organic species, such as anomeric carbons of carbohydrates, conjugated double bonds of aliphatic acids, CRAM, N-acetyls, etc. 1H- 13C heteronuclear single quantum coherence (HSQC) spectrum provides correlation between the aliphatic carbon and its attached protons (Figure. 1).
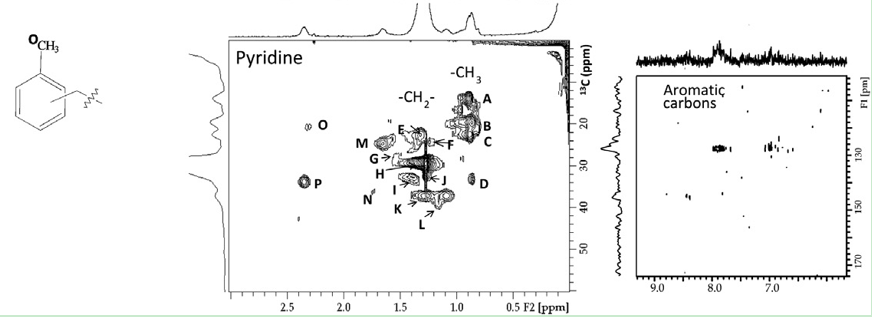 Figure 1. Two-dimensional 1H- 13C HSQC NMR spectra of pyridine.
Figure 1. Two-dimensional 1H- 13C HSQC NMR spectra of pyridine.
Solid-state NMR is nowadays a well-established technique for structure elucidation. It allows investigation on short-range ordering (local environment) and structure. Several techniques have been developed for handling heteronuclear dipolar, homonuclear dipolar, and quadrupolar interactions (Table 1).
Table 1. Techniques used in high-resolution solid-state NMR.
| Techniques | Handling of the following interactions/phenomena |
| Dipolar decoupling (DD) | Heteronuclear dipolar interactions |
| Multiple pulse sequences (MPS) | Homonuclear dipolar interactions |
| Magic angle spinning (MAS) | Chemical shift anisotropy (CSA), dipolar and first-order quadrupolar interactions |
| Dynamic angle spinning (DAS) | Second-order quadrupolar interactions |
| Double orientation rotation (DOR) or multiple quantum NMR cross polarization (CP) | Long spin-lattice relaxation times |
One example of MAS is cross-polarization magic-angle spinning 13C NMR (CPMAS 13C NMR). Analysis performed with variable contact-time pulse sequences showed that the fractions contained aromatic, alkyl, and carbohydrate-like components. CPMAS 13C NMR has been increasingly used to identify the chemical composition of complex mixtures, natural organic matter for instance, and to quantify the distribution of different carbon nuclei by application of specific pulse sequences. CPMAS 13C NMR spectroscopy has also been recognized for its potential in providing conformational information about organic matter by usage of special pulse sequences designed to measure relaxation times.
Our NMR team at Creative Biostructure has extensive experience with molecular characterization and structure elucidation of both small organic molecules and biomacromolecules. Our specialists collaborate with customers to accelerate the research progress. Please contact us for detailed inquiry!
Ordering Process
References
- Nebbioso A. and Piccolo A., (2013). Molecular characterization of dissolved organic matter (DOM): a critical review. Anal Bioanal Chem. 405: 109-124.
- Salmon E., Behar F., and Hatcher P., (2011). Molecular characterization of type 1kerogen from the green river formation using advanced NMR techniques in combination with electrospray ionization/ultrahigh resolution mass spectrometry. Organic Geochemistry. 42: 301-315.
- Stocker M., (2001). Product characterization by NMR. Verified Syntheses of Zeolitic materials. 57-61.
