Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful analytical tool for studying the structure, dynamics, and interactions of molecules. While solution-state NMR is commonly used for the analysis of liquids and dissolved samples, solid-state NMR (ssNMR) is specifically designed for the study of solids, including crystalline materials, amorphous substances, and biological samples such as membrane proteins and fibrils. ssNMR has become an essential technique in materials science, chemistry and biophysics, providing insights that other methods cannot.
Creative Biostructure offers comprehensive ssNMR services that provide information on the structural symmetry, dynamics and interactions of molecules and complexes, which are well suited for studying the microstructure and kinetic behavior of various types of amorphous solid materials.
Fundamentals of Solid-State NMR
The Basics of NMR Spectroscopy
NMR spectroscopy is based on the interaction of atomic nuclei with an external magnetic field. Certain nuclei, such as 1H, 13C, 15N, and 31P, possess a property called nuclear spin that allows them to absorb and emit electromagnetic radiation when exposed to a strong magnetic field. The frequency of this absorption, called the Larmor frequency, depends on the strength of the external magnetic field and the local electronic environment around the nucleus.
In solution-state NMR, rapid molecular motion averages out anisotropic (directional) interactions, resulting in narrow, well-resolved peaks. In solids, however, molecular motion is restricted, resulting in broad, complex spectra due to strong dipolar couplings, chemical shift anisotropy (CSA), and quadrupolar interactions. To overcome these challenges, solid-state NMR employs specialized techniques such as magic angle spinning (MAS) and cross polarization (CP).
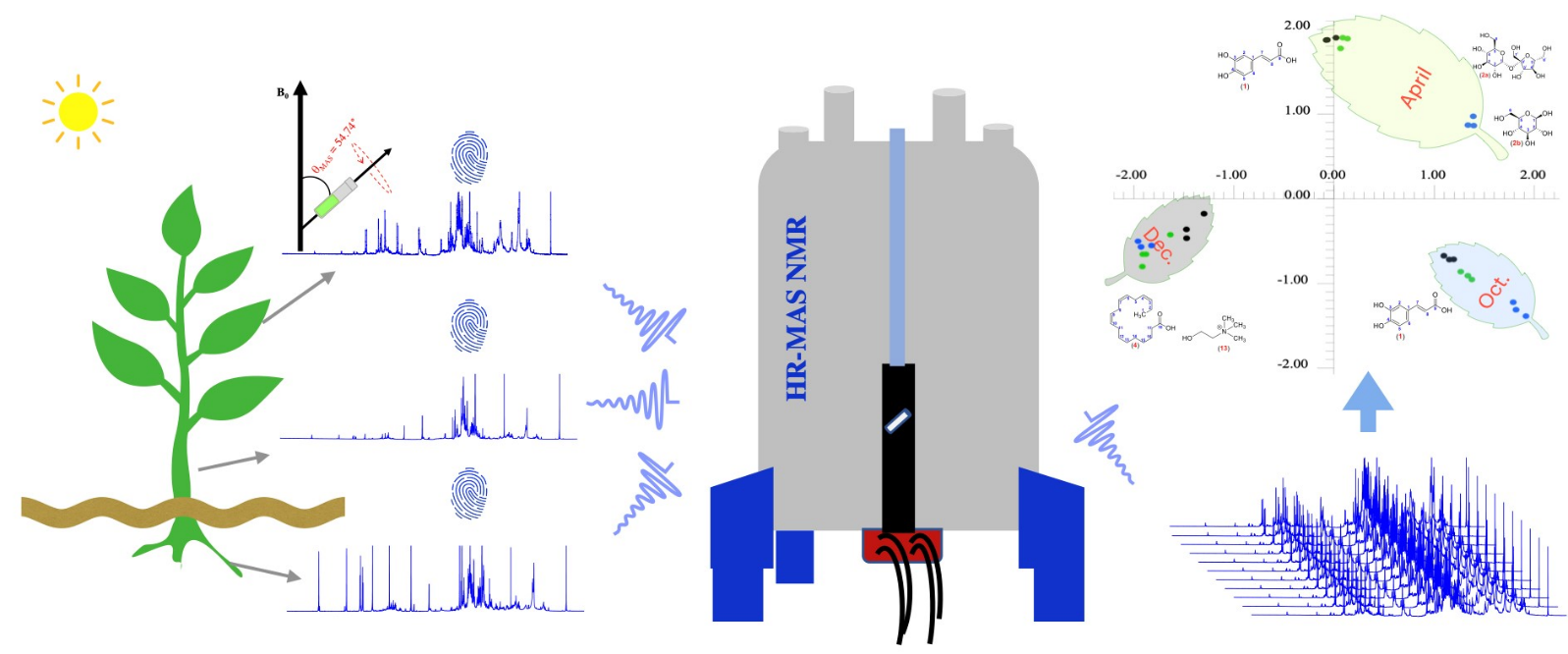
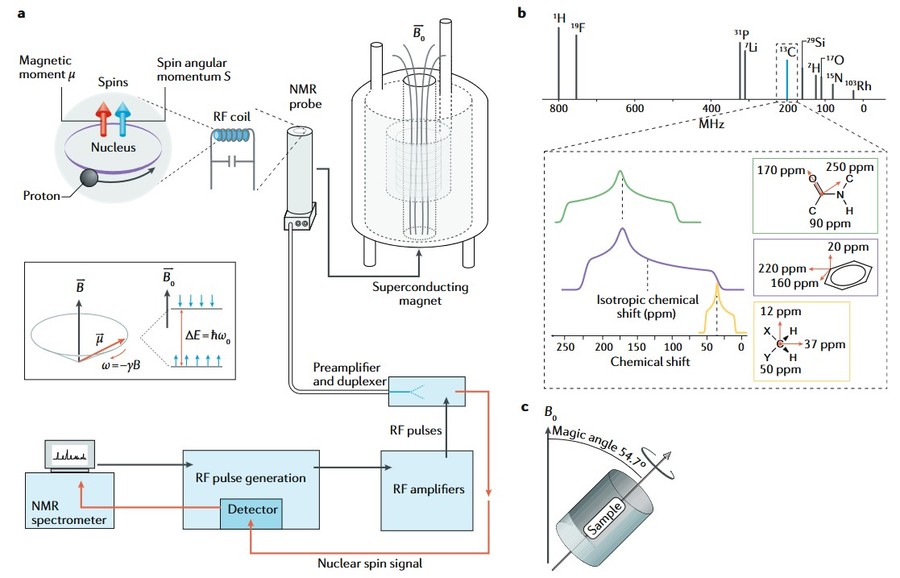 Figure 1. Basics of solid-state NMR spectroscopy for structural analysis of biomolecules and materials. a | Nuclear spin magnetic dipole moments (μ) process around a static magnetic field (B0) at a frequency identical to the transition frequency between the energy levels of the spins (ΔE = ħω0). A radiofrequency (RF) coil is wrapped around the sample at the top of a nuclear magnetic resonance (NMR) probe, which is inserted into the center of the magnet. The RF coil allows irradiation of the RF pulses as well as detection of the transition frequency of the nuclear magnetic moment. Angular velocity, ω = −γB. b | The NMR frequencies of different nuclear isotopes depend on their gyromagnetic ratios (γ) and the magnetic field. In addition, for spins of the same isotope, the frequency depends on the electronic environment of the individual nuclei. Schematic NMR spectra of a static powder containing three 13C nuclei report the chemical structure of the functional groups. The broad powder pattern reflects chemical shift anisotropy, whose geometric average corresponds to the isotropic chemical shift, which is detected when the sample undergoes magic-angle spinning (MAS). c | MAS of the sample in the rotor yields high- resolution NMR spectra of solids by averaging the anisotropic part of the interaction to zero. ppm, parts per million. (Reif et al., 2021)
Figure 1. Basics of solid-state NMR spectroscopy for structural analysis of biomolecules and materials. a | Nuclear spin magnetic dipole moments (μ) process around a static magnetic field (B0) at a frequency identical to the transition frequency between the energy levels of the spins (ΔE = ħω0). A radiofrequency (RF) coil is wrapped around the sample at the top of a nuclear magnetic resonance (NMR) probe, which is inserted into the center of the magnet. The RF coil allows irradiation of the RF pulses as well as detection of the transition frequency of the nuclear magnetic moment. Angular velocity, ω = −γB. b | The NMR frequencies of different nuclear isotopes depend on their gyromagnetic ratios (γ) and the magnetic field. In addition, for spins of the same isotope, the frequency depends on the electronic environment of the individual nuclei. Schematic NMR spectra of a static powder containing three 13C nuclei report the chemical structure of the functional groups. The broad powder pattern reflects chemical shift anisotropy, whose geometric average corresponds to the isotropic chemical shift, which is detected when the sample undergoes magic-angle spinning (MAS). c | MAS of the sample in the rotor yields high- resolution NMR spectra of solids by averaging the anisotropic part of the interaction to zero. ppm, parts per million. (Reif et al., 2021)
Key Techniques in Solid-State NMR
Magic Angle Spinning (MAS)
One of the major advances in ssNMR is Magic Angle Spinning (MAS), a technique that reduces dipolar interactions and chemical shift anisotropy by rapidly spinning the sample at a precise angle of 54.74° relative to the external magnetic field. This angle is called the magic angle because it minimizes the second-order anisotropic interactions that cause peak broadening.
- High-speed MAS (≥60 kHz) can fully average out dipolar interactions, improving resolution.
- Moderate-speed MAS (~10–30 kHz) is often used in conjunction with cross-polarization and decoupling.
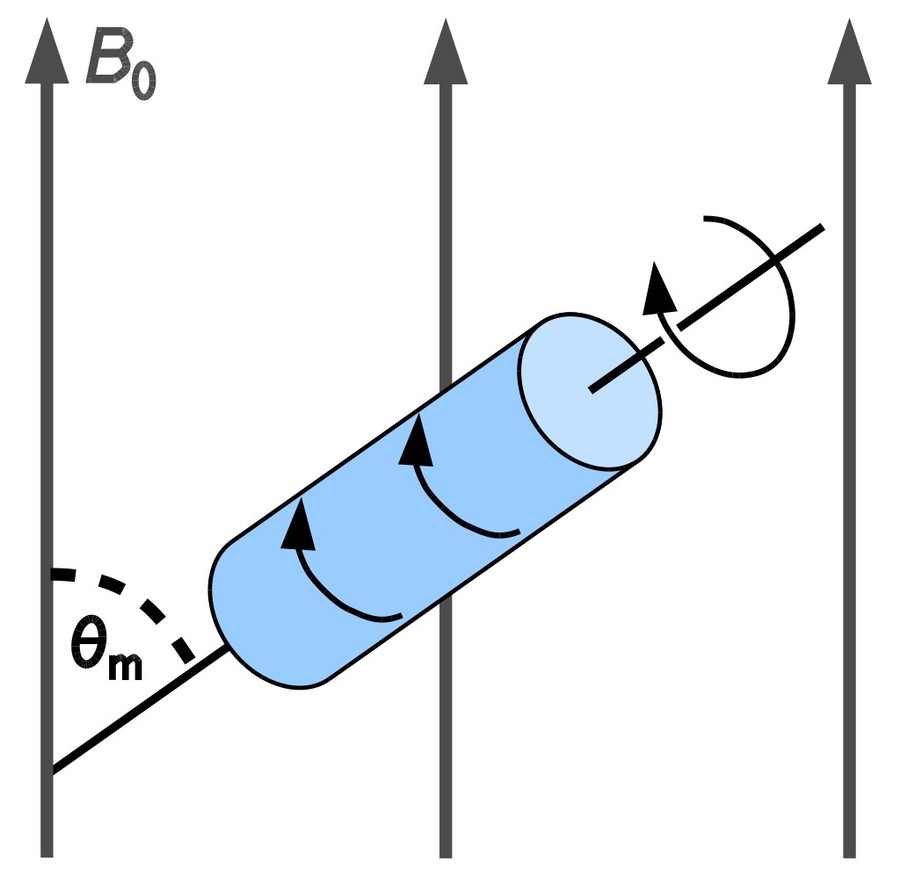 Figure 2. Magic-angle spinning: The sample (blue) is rotating with high frequency inside the main magnetic field (B0). The axis of rotation is tilted by the magic angle θm with respect to the direction of B0.
Figure 2. Magic-angle spinning: The sample (blue) is rotating with high frequency inside the main magnetic field (B0). The axis of rotation is tilted by the magic angle θm with respect to the direction of B0.
Cross-Polarization (CP) and Decoupling
Since some nuclei (e.g., 13C and 15N) have low natural abundance, their direct detection is inefficient. Cross-polarization (CP) enhances signal sensitivity by transferring polarization from abundant spins (e.g., 1H) to less sensitive nuclei (e.g., 13C).
In addition, heteronuclear decoupling is used to remove dipolar couplings between different types of nuclei (e.g., 1H-13C), resulting in sharper peaks. Decoupling techniques such as SPINAL-64 and TPPM (Two-Pulse Phase Modulation) are commonly used in MAS experiments.
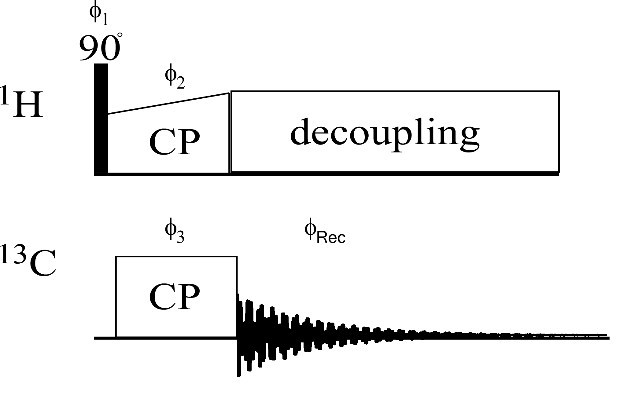 Figure 3. Cross-polarization (CP) pulse sequence. The phases of the proton CP pulse and excitation pulse are perpendicular in order to lock the magnetization. The complete phase cycle is as follows (in degrees): ϕ1 = 0, 180; ϕ2 = 90; ϕ3 = 270, 270, 180, 180; ϕrec = 180, 0, 90, 270. (Morag et al., 2018)
Figure 3. Cross-polarization (CP) pulse sequence. The phases of the proton CP pulse and excitation pulse are perpendicular in order to lock the magnetization. The complete phase cycle is as follows (in degrees): ϕ1 = 0, 180; ϕ2 = 90; ϕ3 = 270, 270, 180, 180; ϕrec = 180, 0, 90, 270. (Morag et al., 2018)
Recoupling and Dipolar-Based Methods
While MAS averages out anisotropic interactions, sometimes these interactions provide useful structural information. Recoupling techniques such as REDOR (Rotational-Echo Double Resonance) and TEDOR (Transferred-Echo Double Resonance) selectively reintroduce dipolar interactions to measure distances between nuclei, which is valuable for the study of biomolecules and materials.
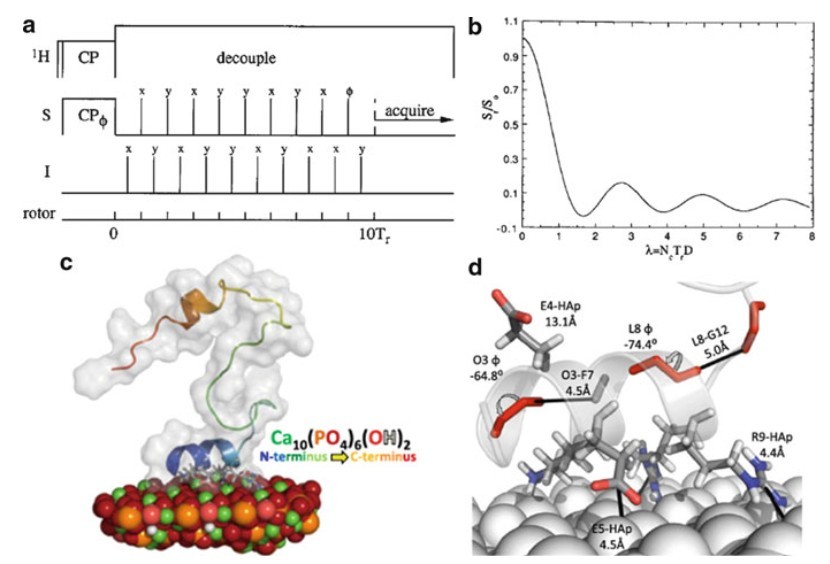 Figure 4. (a) REDOR experiment and (b) typical REDOR dephasing curve (D designates the dipole–dipole coupling, τr the rotor period, and Nc the number of rotor cycles for the REDOR experiment). (c, d) REDOR based determination of statherin binding to a hydroxyapatite surface (Nielsen et al., 2011)
Figure 4. (a) REDOR experiment and (b) typical REDOR dephasing curve (D designates the dipole–dipole coupling, τr the rotor period, and Nc the number of rotor cycles for the REDOR experiment). (c, d) REDOR based determination of statherin binding to a hydroxyapatite surface (Nielsen et al., 2011)
Quadrupolar NMR
Many nuclei (e.g., 27Al, 23Na, 17O) have quadrupolar moments, meaning they interact strongly with electric field gradients in solids, leading to broad, complex spectra. Multiple-quantum MAS (MQMAS) and Double-Quantum Filtering (DQF) help resolve these spectra, enabling structural insights into materials like zeolites, ceramics, and glasses.
Differences Between Solution-State and Solid-State NMR
Solution-state NMR (Nuclear Magnetic Resonance) is a powerful spectroscopic technique used to study molecular structures, dynamics, and interactions in solution. By analyzing how atomic nuclei respond to a magnetic field, it provides detailed information about chemical environments, connectivity, and conformational changes. Widely applied in chemistry, biochemistry, and drug discovery, solution-state NMR is particularly valuable for characterizing small molecules, proteins, and nucleic acids in their native, liquid-phase conditions. The differences between solution-state NMR and solid-state NMR are summarized in the table below.
| Feature | Solution-State NMR | Solid-State NMR |
|---|---|---|
| Sample State | Liquid | Solid |
| Molecular Motion | Rapid | Restricted |
| Peak Width | Narrow | Broad |
| Signal Resolution | High | Lower (without MAS) |
| Experimental Techniques | 1D and 2D spectroscopy | MAS, CP, Decoupling |
| Applications | Small molecules, proteins, and metabolites | Crystalline and amorphous solids, materials, polymers, proteins |
Applications of Solid-State NMR
Structural Biology: Proteins and Membranes
ssNMR is a critical tool in the study of proteins and biomolecular structures that are difficult to analyze using traditional methods such as X-ray crystallography or solution NMR. ssNMR excels in the analysis of complex, rigid or dynamic structures, making it ideal for
- Membrane Proteins: These proteins are embedded in lipid bilayers and are notoriously difficult to study with conventional NMR due to their hydrophobic nature and size. ssNMR provides high-resolution insight into the structure and function of these proteins, which are essential for many biological processes such as signal transduction and transport across membranes.
- Amyloid Fibrils: ssNMR is a powerful technique for studying amyloid fibrils, protein aggregates associated with neurodegenerative diseases such as Alzheimer's, Parkinson's and Huntington's. By resolving the atomic-level structure of amyloid fibrils, ssNMR provides insight into their formation and the mechanisms underlying disease progression, offering a potential avenue for therapeutic intervention.
- Protein-Ligand Interactions: Understanding the binding interactions between proteins and small molecules is critical to drug discovery and development. ssNMR allows researchers to study these interactions in the solid state, providing a more realistic view of how ligands bind to their protein targets under biological conditions, which can guide the design of more effective drugs.
Materials Science and Polymers
ssNMR plays a vital role in materials science by providing atomic-level insights into the structure and behavior of various materials. It is especially valuable in:
- Polymers and Composites: ssNMR allows the study of the molecular mobility, chain dynamics and cross-linking behavior of polymers. This is particularly useful for designing new polymeric materials with enhanced properties such as strength, flexibility, or thermal stability. ssNMR can also reveal how polymers interact with other materials in composite structures, providing insight into the performance and durability of these materials in real-world applications.
- Battery Materials: ssNMR is indispensable in the study of battery materials, especially for lithium-ion batteries, where it helps to analyze the atomic structure of electrode materials. For example, 7Li, 19F, and 31P ssNMR can be used to study the behavior of lithium ions, fluorine, and phosphorus atoms in the electrodes, providing valuable information on ion diffusion, intercalation mechanisms, and the stability of battery components, all of which are critical to improving battery efficiency and lifetime.
- Zeolites and Catalysts: In catalysis, ssNMR provides detailed insight into the atomic arrangement of zeolites and metal sites within catalytic materials. By studying the framework structures and interactions at the metal sites, ssNMR helps to design more efficient catalysts for industrial processes such as petroleum refining and chemical synthesis, contributing to advances in energy production and environmental sustainability.
Pharmaceutical Industry
In the pharmaceutical industry, ssNMR is an essential tool for understanding the molecular characteristics of drugs and their formulations. The ability to analyze both crystalline and amorphous phases enables ssNMR to:
- Characterizing Polymorphs: Polymorphism, the ability of a compound to exist in multiple crystal forms, can significantly affect the solubility, stability and bioavailability of a drug. ssNMR helps to characterize the different polymorphs of active pharmaceutical ingredients (APIs), which is critical for optimizing drug formulation and ensuring consistent therapeutic efficacy.
- Detecting Amorphous Content: The amorphous form of drugs, which lacks a defined crystal structure, is often more soluble but less stable than crystalline forms. ssNMR is highly effective in quantifying the amorphous content of drug formulations, enabling pharmaceutical companies to optimize the stability and shelf life of drugs.
- Studying API-Excipient Interactions: The interaction between APIs and excipients (inactive ingredients) can have a significant impact on drug formulation, influencing factors such as drug release rate, bioavailability and stability. ssNMR allows the detailed study of these interactions at the molecular level, facilitating the development of more effective and reliable drug delivery systems.
Geochemistry and Earth Sciences
In geochemistry and earth sciences, ssNMR is used to gain deeper insight into the composition and behavior of minerals and materials, providing valuable information for a wide range of applications:
- Silicates and Minerals: ssNMR is used extensively to study the atomic structures of silicates and minerals, particularly through 29Si and 27Al NMR. These studies help scientists understand the formation, stability and chemical reactivity of minerals in different geological environments, including their role in processes such as weathering and soil formation.
- Cement Chemistry: ssNMR plays a crucial role in cement chemistry, helping to analyze the hydration and setting processes that occur when cement interacts with water. By studying the atomic-level structure of cement phases during these processes, ssNMR provides insight into the strength development and long-term durability of the material, which is essential for optimizing cement formulations for construction and infrastructure projects.
- Glasses and Ceramics: ssNMR is used to probe the atomic structures of glasses and ceramics, revealing their fundamental properties. This is particularly important for the design of new materials with specific properties such as increased heat resistance, mechanical strength or electrical conductivity, which have broad applications in industries ranging from electronics to aerospace.
Advantages and Limitations of Solid-State NMR
| Advantages | Limitations |
|---|---|
| Non-destructive: Does not alter the sample's structure. | Lower sensitivity: Compared to solution NMR, requiring signal enhancement methods. |
| Applicable to non-crystalline materials: Unlike X-ray crystallography. | Broad spectra: Without MAS, peaks can be too broad to interpret. |
| Provides atomic-level insights: Without requiring large, pure crystals. | Long experiment times: Due to weak signals and slow data acquisition. |
| Can study complex, heterogeneous systems: e.g., biological membranes, polymers, and pharmaceuticals. | Expensive instrumentation: High-field magnets and MAS probes are costly. |
Case Study
Case 1: Advanced Solid-State NMR Techniques for Characterization of Membrane Protein Structure and Dynamics: Application to Anabaena Sensory Rhodopsin
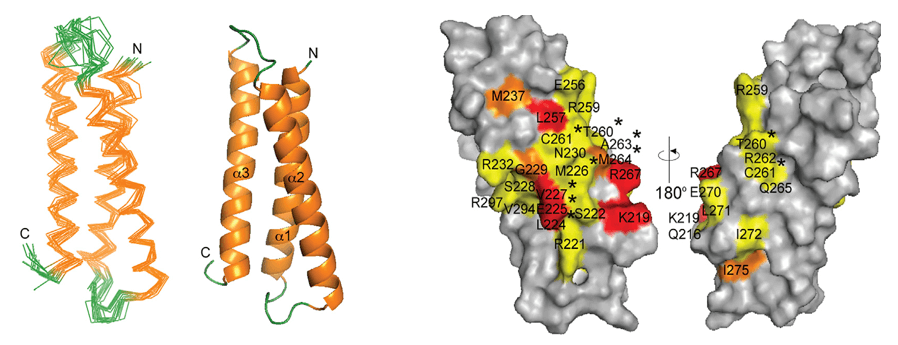
The study of membrane proteins (MPs) in terms of their structure, dynamics and function has long been a central application of solid-state NMR (ssNMR). Recent advances in instrumentation, coupled with the development of numerous new ssNMR techniques over the past decade, have enabled the determination of high-resolution structures and models for both bitopic and polytopic α-helical MPs. However, challenges remain due to the need to preserve lipids in the sample, the dominance of a single secondary structure type, variable dynamics, and the potential for local disorder in the loop regions, all of which complicate structure determination. In this perspective, the researchers discuss their recent work on determining the structure and functional dynamics of Anabaena sensory rhodopsin (ASR), a heptahelical transmembrane (7TM) protein. They also review both established and emerging ssNMR methods applicable to structure determination, with a focus on techniques used to study ASR, a bacterial protein that shares its 7TM architecture with G protein-coupled receptors.
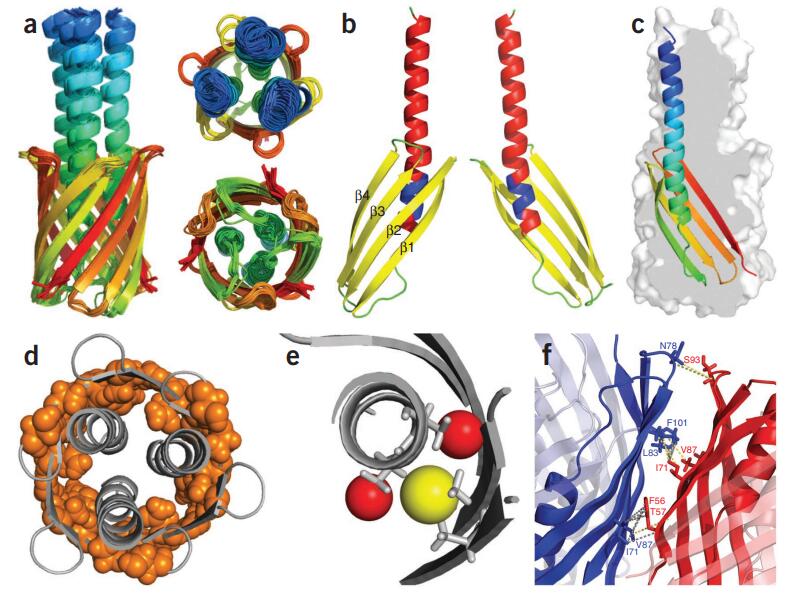
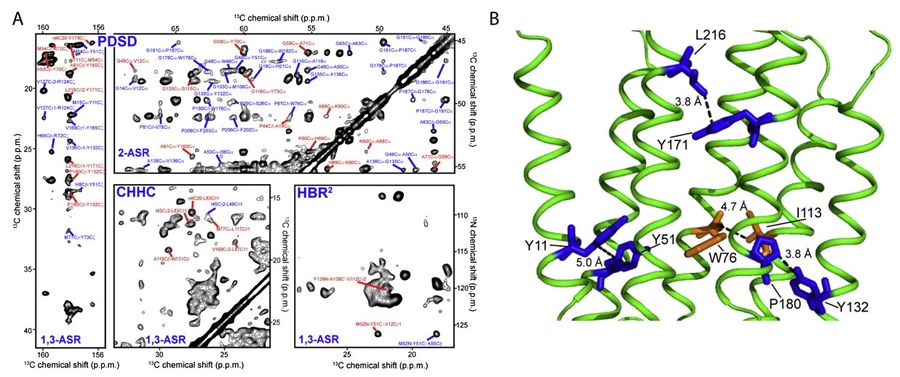 Figure 5. (A) Examples of PDSD, CHHC and HBR2 ssNMR spectra of ASR recorded on sparsely labeled samples (1,3-ASR, 2-ASR, as indicated in the figures). The HBR2 spectrum is a 2D NC plane extracted from the 3D experiment at the carbonyl shift of 178.2 ppm. In all spectra, cross-peaks corresponding to intrahelical restraints and restraints in the loops connecting adjacent helices (excluding the b-stranded B–C loop) are labeled in blue while interhelical correlations, and correlations defining the short b-hairpin within the B–C loop are labeled in red. (B) Several of the distance restraints used for the initial structural template calculation visualized on the ASR structure. The residues involved in connectivities obtained from the PDSD spectra are shown in blue, while those obtained from HBR2 data are shown in orange. The cytoplasmic side is on top. (Ward et al., 2014)
Figure 5. (A) Examples of PDSD, CHHC and HBR2 ssNMR spectra of ASR recorded on sparsely labeled samples (1,3-ASR, 2-ASR, as indicated in the figures). The HBR2 spectrum is a 2D NC plane extracted from the 3D experiment at the carbonyl shift of 178.2 ppm. In all spectra, cross-peaks corresponding to intrahelical restraints and restraints in the loops connecting adjacent helices (excluding the b-stranded B–C loop) are labeled in blue while interhelical correlations, and correlations defining the short b-hairpin within the B–C loop are labeled in red. (B) Several of the distance restraints used for the initial structural template calculation visualized on the ASR structure. The residues involved in connectivities obtained from the PDSD spectra are shown in blue, while those obtained from HBR2 data are shown in orange. The cytoplasmic side is on top. (Ward et al., 2014)
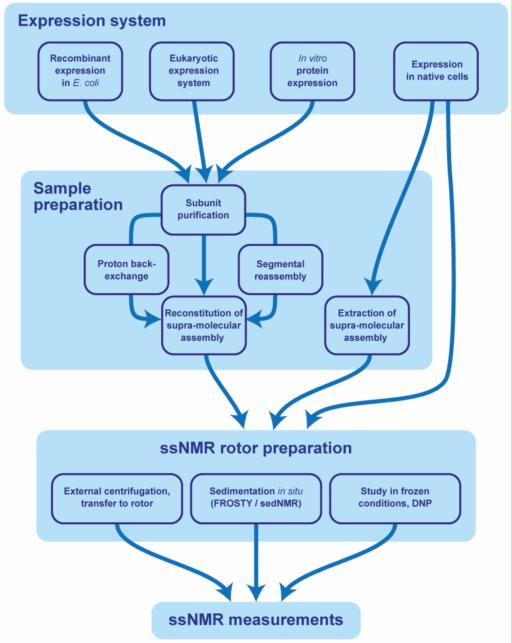
Case 2: Sedimentation Yields Long-Term Stable Protein Samples as Shown by Solid-State NMR
Currently, sedimentation of proteins in a magic angle spinning (MAS) rotor provides a fast and reliable method for solid-state nuclear magnetic resonance (NMR) sample preparation, allowing the study of a range of non-crystalline protein samples. Protein concentrations of up to 400 mg/mL can be achieved, with the protein constituting approximately 50-60% of the NMR rotor content, while the remainder consists of a buffer solution containing counterions to balance the charge of the protein. In this study, the researchers demonstrate the long-term stability of four sedimented proteins and their complexes with nucleotides, including a bacterial DnaB helicase, an ABC transporter, an archaeal primase, and an RNA polymerase subunit. Solid-state NMR spectra obtained immediately after sample preparation and up to five years later showed no significant differences or loss of signal intensity, indicating that protein sediments in the rotor can remain stable for long periods of time. Using an ABC transporter as an example, we further demonstrate that the protein not only retains its structure but also its functionality after long-term storage in the sedimented state.
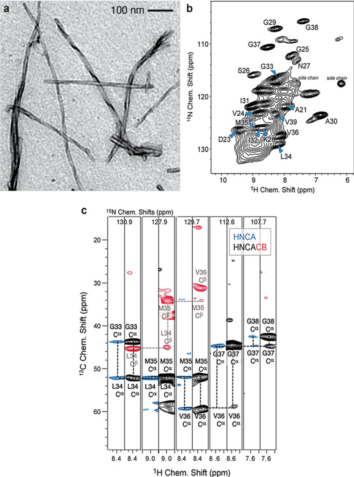
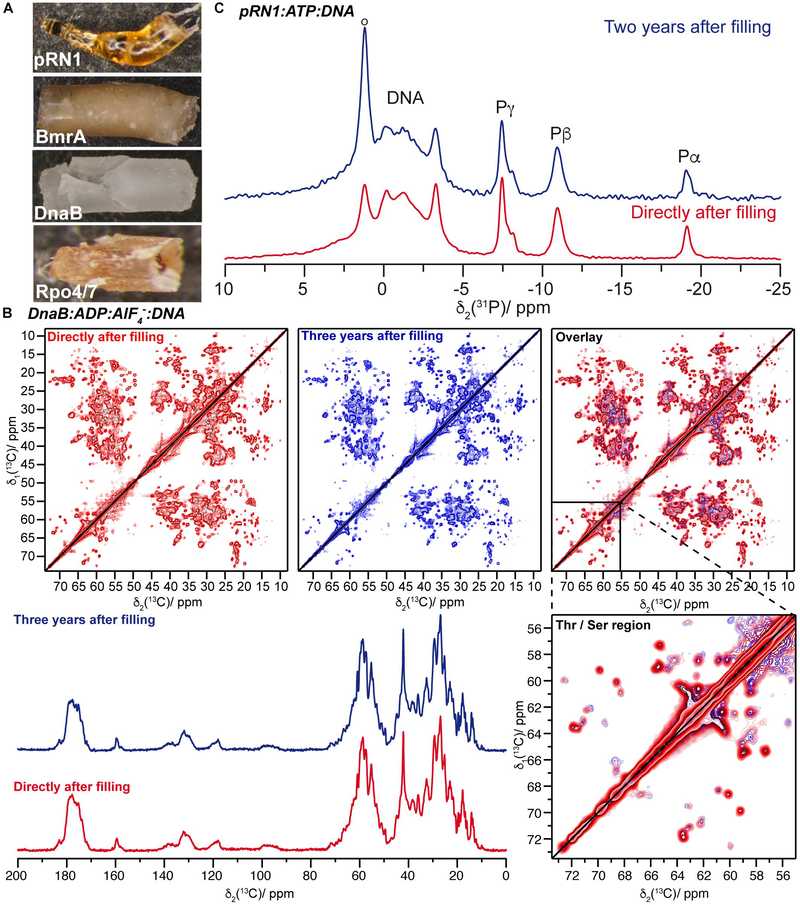 Figure 6. (A) Protein sediments drilled out of solid-state NMR rotors of Rpo4/7+, DnaB, BmrA, and the pRN1 primase (bottom to top). These sediments belong to different protein samples and not systematically to the ones for which spectra are reported herein. (B) The top panel shows the comparison of 20 ms 13C-13C DARR correlation spectra of DnaB:ADP:AlF4–:DNA recorded directly after rotor filling (red) and 3 years later (blue). The bottom panel presents a 13C,1H 1D CP MAS spectra recorded on DnaB:ADP:AlF4–:DNA. (C) 31P,1H CPMAS spectra of the quaternary pRN1:ATP:ATP:DNA complex collected directly after sample filling (red) and 2 years later (blue). (Wiegand et al., 2020)
Figure 6. (A) Protein sediments drilled out of solid-state NMR rotors of Rpo4/7+, DnaB, BmrA, and the pRN1 primase (bottom to top). These sediments belong to different protein samples and not systematically to the ones for which spectra are reported herein. (B) The top panel shows the comparison of 20 ms 13C-13C DARR correlation spectra of DnaB:ADP:AlF4–:DNA recorded directly after rotor filling (red) and 3 years later (blue). The bottom panel presents a 13C,1H 1D CP MAS spectra recorded on DnaB:ADP:AlF4–:DNA. (C) 31P,1H CPMAS spectra of the quaternary pRN1:ATP:ATP:DNA complex collected directly after sample filling (red) and 2 years later (blue). (Wiegand et al., 2020)
Creative Biostructure has extensive experience in using ssNMR to analyze the fundamental chemical structure, spatial arrangement, and conformational properties of a wide range of materials, including organic and inorganic compounds, polymers, natural products, small molecule proteins, and more. In addition to helping our clients obtain structural details such as chemical shifts and J-coupling, we also provide insight into the relative positions of specific atoms within a sample, including interatomic distances and orientations. Contact us today to discuss your project!
References
- Morag O, Sgourakis NG, Abramov G, Goldbourt A. Filamentous bacteriophage viruses: preparation, magic-angle spinning solid-state NMR experiments, and structure determination. In: Ghose R, ed. Protein NMR. Vol 1688. Springer New York; 2018:67-97. doi:10.1007/978-1-4939-7386-6_4
- Nielsen NC, Strass LA, Nielsen AB. Dipolar recoupling. In: Chan JCC, ed. Solid State NMR. Vol 306. Springer Berlin Heidelberg; 2011:1-45. doi:10.1007/128_2011_129
- Page SJ, Gallo A, Brown SP, Lewandowski JR, Hanna JV, Franks WT. Simultaneous MQMAS NMR experiments for two half-integer quadrupolar nuclei. Journal of Magnetic Resonance. 2020;320:106831. doi:10.1016/j.jmr.2020.106831
- Reif B, Ashbrook SE, Emsley L, Hong M. Solid-state NMR spectroscopy. Nat Rev Methods Primers. 2021;1(1):2. doi:10.1038/s43586-020-00002-1
- Ward ME, Brown LS, Ladizhansky V. Advanced solid-state NMR techniques for characterization of membrane protein structure and dynamics: Application to Anabaena Sensory Rhodopsin. Journal of Magnetic Resonance. 2015;253:119-128. doi:10.1016/j.jmr.2014.11.017
- Wiegand T, Lacabanne D, Torosyan A, et al. Sedimentation yields long-term stable protein samples as shown by solid-state NMR. Front Mol Biosci. 2020;7. doi:10.3389/fmolb.2020.00017