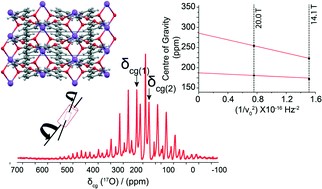
Nuclear Magnetic Resonance (NMR) spectroscopy has long been a cornerstone in the elucidation of molecular structure, dynamics, and interactions. Since its inception, NMR has evolved, incorporating technological advances that have greatly improved its sensitivity, resolution, and applicability to various scientific disciplines. This comprehensive resource delves into the latest advances in NMR technology, exploring innovations that are redefining the landscape of molecular spectroscopy.
At Creative Biostructure, we invite you to explore the latest advances in Nuclear Magnetic Resonance (NMR) technology. Utilizing our state-of-the-art NMR platform, we offer a comprehensive suite of advanced NMR services designed to meet the evolving needs of researchers and industry professionals. Our NMR platform is equipped with the latest instrumentation and methodologies, enabling us to provide high-resolution, high-throughput NMR solutions for a wide range of applications. Whether you are involved in structural biology, drug discovery, materials science, or any other field that benefits from NMR analysis, our team of experts is dedicated to providing you with unparalleled support and innovative solutions.
 Figure 1. Advanced NMR techniques.
Figure 1. Advanced NMR techniques.
Solid-State NMR Spectroscopy
Solid-state NMR (ssNMR) spectroscopy has undergone remarkable transformations, enabling detailed investigations of insoluble and non-crystalline materials, including complex biomolecules and advanced materials.
Rotational Echo Double Resonance (REDOR)
REDOR is a key technique in ssNMR that facilitates the measurement of heteronuclear dipolar couplings by reintroducing interactions averaged out by Magic Angle Spinning (MAS). This method compares the intensity of NMR signals with and without dephasing pulses, allowing precise distance measurements between different types of nuclei. REDOR has become an essential tool in NMR crystallography studies, providing insight into molecular structures that are difficult to crystallize.
Ultra-Fast Magic Angle Spinning (MAS) for Proton NMR
Proton (1H) NMR in solids has historically been hampered by strong homonuclear dipolar interactions, resulting in broad NMR lines and short transverse relaxation times (T2). The advent of ultrafast MAS techniques has mitigated these challenges by more effectively averaging out dipolar couplings, resulting in sharper NMR lines. This improvement has made proton-detected ssNMR as versatile as solution-state NMR, enabling high-resolution multidimensional experiments and providing structurally valuable constraints essential for studying material dynamics.
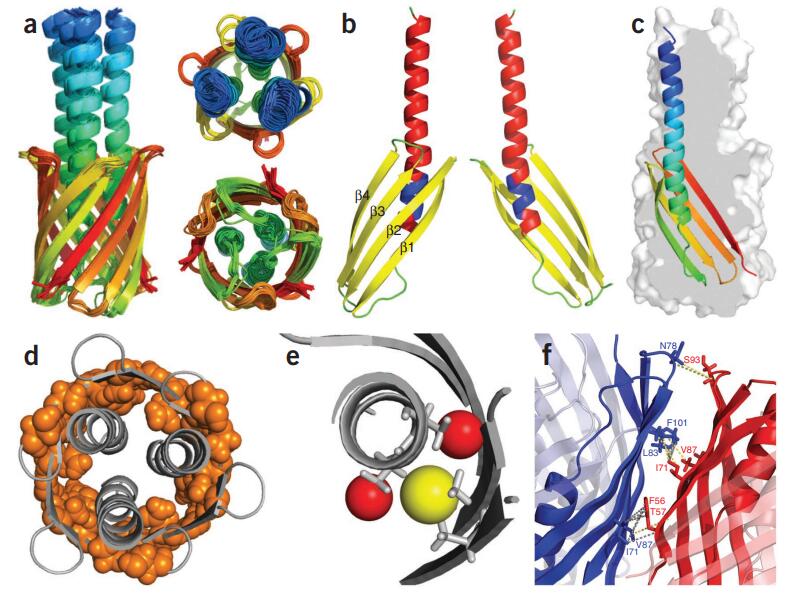
Case Study: Three Decades of REDOR in Protein Science: A Solid-State NMR Technique for Distance Measurement and Spectral Editing
Solid-state NMR (ssNMR) is a powerful tool to investigate non-crystallizable, poorly soluble molecular systems, such as membrane proteins, amyloids, and cell walls, in environments that closely resemble their physical sites of action. Rotational-echo double resonance (REDOR) is an ss-NMR methodology, which by reintroducing heteronuclear dipolar coupling under magic angle spinning conditions provides intramolecular and intermolecular distance restraints at the atomic level.
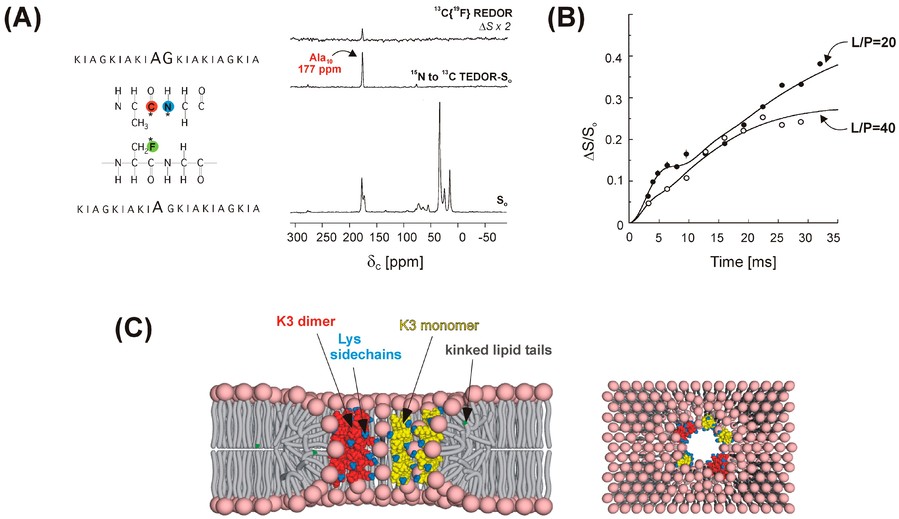 Figure 2. Dipolar recoupling for spectral editing and intermolecular distance restraints. (A) Positions of 13C, 15N, and 19F labels in two antimicrobial (KIAGKIA)3 (K3) peptide analogues used in REDOR experiments to probe the secondary structure and aggregation properties (left). The 15N→13C{19F} TEDOR-REDOR NMR spectra of a 1:1 mixture of [1-13C]Ala10-[15N]Gly11-K3-NH2 and [3-19F]Ala10-K3-NH2 incorporated into multilamellar phospholipid vesicles (right). (B) The 13C{19F} REDOR dephasing of a 1:1 mixture of peptides in (A) at lipid-to-peptide molar ratios of L/P = 20 and L/P = 40. (C) REDOR-derived model of the K3 membrane pore: cross-section (left) and top view (right). K3 monomers and dimers are shown in yellow and red, respectively. Lysine side chains are indicated in blue. Peptide chains and lipid headgroups together line the wall of the torus-like pore. (Toke, 2023)
Figure 2. Dipolar recoupling for spectral editing and intermolecular distance restraints. (A) Positions of 13C, 15N, and 19F labels in two antimicrobial (KIAGKIA)3 (K3) peptide analogues used in REDOR experiments to probe the secondary structure and aggregation properties (left). The 15N→13C{19F} TEDOR-REDOR NMR spectra of a 1:1 mixture of [1-13C]Ala10-[15N]Gly11-K3-NH2 and [3-19F]Ala10-K3-NH2 incorporated into multilamellar phospholipid vesicles (right). (B) The 13C{19F} REDOR dephasing of a 1:1 mixture of peptides in (A) at lipid-to-peptide molar ratios of L/P = 20 and L/P = 40. (C) REDOR-derived model of the K3 membrane pore: cross-section (left) and top view (right). K3 monomers and dimers are shown in yellow and red, respectively. Lysine side chains are indicated in blue. Peptide chains and lipid headgroups together line the wall of the torus-like pore. (Toke, 2023)
Dynamic Nuclear Polarization (DNP)
Dynamic nuclear polarization (DNP) has emerged as a transformative technique for enhancing NMR sensitivity by transferring the polarization of unpaired electrons to nearby nuclei. This process utilizes the higher electron spin polarization achieved by microwave irradiation near electron paramagnetic resonance (EPR) transitions to enhance nuclear spin polarization. The result is a significant enhancement of NMR signal intensities, facilitating the study of samples with low concentrations or limited volumes.
Mechanisms of DNP
DNP operates by several mechanisms, including the Overhauser Effect (OE), Solid Effect (SE), Cross Effect (CE), and Thermal Mixing (TM). Each mechanism involves different interactions between electron and nuclear spins that determine the efficiency and applicability of DNP under different conditions. For example, OE is dominant in solution-phase NMR, while SE, CE, and TM are more relevant in solid-state NMR applications.
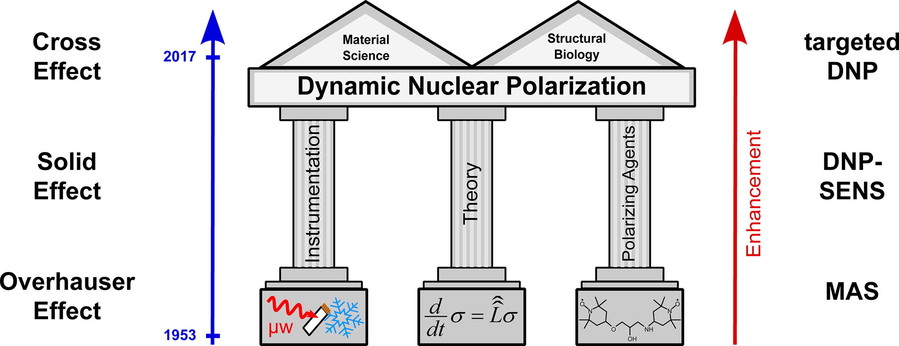 Figure 3. Overview of dynamic nuclear polarization: development, theory and applications. (Thankamony et al., 2017)
Figure 3. Overview of dynamic nuclear polarization: development, theory and applications. (Thankamony et al., 2017)
Advancements in Polarizing Agents and Instrumentation
The development of efficient polarizing agents such as TOTAPOL, AMUPOL and TEKPOL has been instrumental in improving the performance of DNP. These agents allow for more effective polarization transfer, resulting in significant signal enhancement. In addition, advances in microwave sources, such as klystrons and gyrotrons, have enabled continuous microwave irradiation at frequencies near the EPR transitions, further optimizing DNP efficiency. These innovations have dramatically shortened experimental times, allowing surface observations, insensitive isotopes, and multidimensional experiments on low natural abundance nuclei and dilute species.
Beta-Detected Nuclear Magnetic Resonance (β-NMR)
Beta-detected nuclear magnetic resonance (β-NMR) is a specialized technique that combines the principles of both NMR and beta decay to probe local magnetic and electronic environments within materials. This method is particularly useful in fields such as materials science, condensed matter physics, and chemistry.
Principles of β-NMR
In β-NMR, a beam of radioactive nuclei, such as 8Li or 31Mg, is polarized by optical pumping before being injected into the sample material. The anisotropic beta decay of these nuclei is then monitored to obtain information about the local environment. This technique allows the study of phenomena such as superconductivity, magnetism, and thin film interfaces.
Applications and Facilities
β-NMR has been used to study a wide variety of systems, including high-temperature superconductors and magnetic thin films. Facilities such as TRIUMF in Canada and ISOLDE at CERN provide the necessary infrastructure to conduct β-NMR experiments and provide insight into the behavior of materials at the atomic level.
NMR Crystallography
NMR crystallography is an emerging field that integrates NMR spectroscopy with crystallographic techniques to determine the structures of complex molecules, particularly those that are challenging to crystallize.
NMR Crystallography leverages the sensitivity of NMR to the local atomic environment to provide structural information at the atomic level. Unlike traditional X-ray crystallography, which relies on well-ordered crystals, NMR can be applied to both crystalline and amorphous materials, making it particularly useful for studying systems where crystallization is challenging.
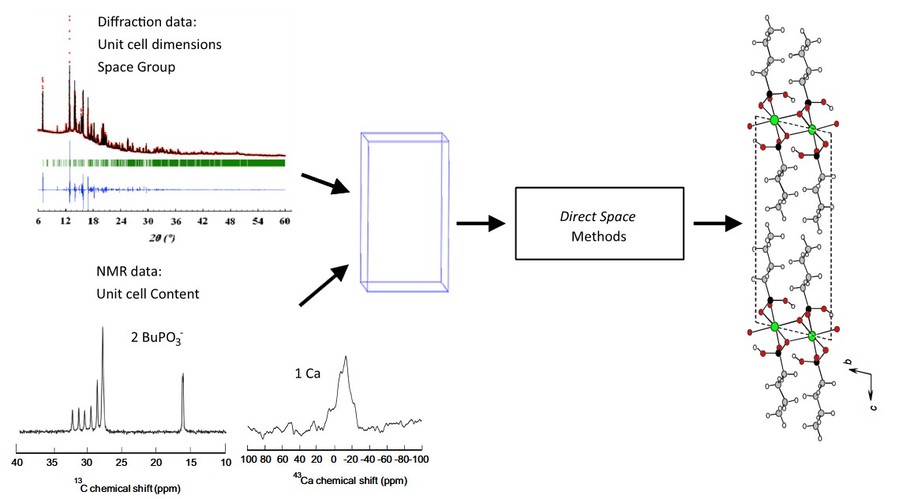 Figure 4. Schematic representation of combining diffraction data (determination of unit cell dimension and space group) and solid-state NMR (determination of the number and nature of the species). The structure search is then performed with a direct space software (FOX in that case), with the powder diffraction diagram as experimental cost function. The example shows the data for Ca(BuPO3). (Martineau, 2014)
Figure 4. Schematic representation of combining diffraction data (determination of unit cell dimension and space group) and solid-state NMR (determination of the number and nature of the species). The structure search is then performed with a direct space software (FOX in that case), with the powder diffraction diagram as experimental cost function. The example shows the data for Ca(BuPO3). (Martineau, 2014)
Integration with Computational Methods
The synergy between NMR data and computational modeling has improved the accuracy of structure determination. By combining experimental NMR parameters with density functional theory (DFT) calculations, researchers can construct detailed models of molecular structures to aid in the design of new materials and pharmaceuticals.
Applications in Drug Development
NMR crystallography has been instrumental in elucidating the structures of pharmaceutical compounds, especially those that do not form crystals suitable for X-ray diffraction. This approach provides critical information about molecular conformations and interactions, facilitating the development of more effective drugs.
Case Study: Refinement of the Crystal Structure of Li4P2S6 Using NMR Crystallography
The crystal structure of Li4P2S6 was determined by a multifaceted approach integrating X-ray powder diffraction, quantum chemical calculations, and solid-state nuclear magnetic resonance (NMR) spectroscopy. Crucial insights into the space group symmetry were obtained from two-dimensional 31P single quantum/double quantum correlation spectra, which facilitated the structure solution using the Rietveld refinement method.
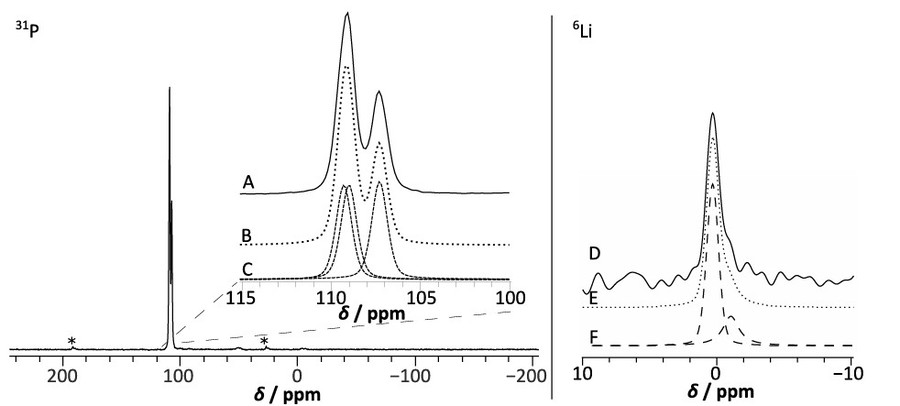 Figure 5. Experimental 31P and 6Li MAS NMR spectra of Li4P2S6 at νrot = 10 kHz. Spinning sidebands are marked with an asterisk. (A) shows the phosphorus peaks of the experimental 31P MAS NMR spectrum of Li4P2S6, (B) shows the simulated 31P spectrum resulting in the simulated peaks with an intensity ratio of 1.0:1.0:1.0 (spectrum (C)). (D) shows the experimental 6Li MAS NMR spectrum of Li4P2S6 at νrot = 10 kHz, (F) shows the 6Li spectrum of two simulated peaks with an peak area ratio of 3.0:1 of spectrum resulting in spectrum (E). Due to the limited resolution of the spectrum, the exact peak area ratio is subject to a bigger uncertainty. (Neuberger et al., 2018)
Figure 5. Experimental 31P and 6Li MAS NMR spectra of Li4P2S6 at νrot = 10 kHz. Spinning sidebands are marked with an asterisk. (A) shows the phosphorus peaks of the experimental 31P MAS NMR spectrum of Li4P2S6, (B) shows the simulated 31P spectrum resulting in the simulated peaks with an intensity ratio of 1.0:1.0:1.0 (spectrum (C)). (D) shows the experimental 6Li MAS NMR spectrum of Li4P2S6 at νrot = 10 kHz, (F) shows the 6Li spectrum of two simulated peaks with an peak area ratio of 3.0:1 of spectrum resulting in spectrum (E). Due to the limited resolution of the spectrum, the exact peak area ratio is subject to a bigger uncertainty. (Neuberger et al., 2018)
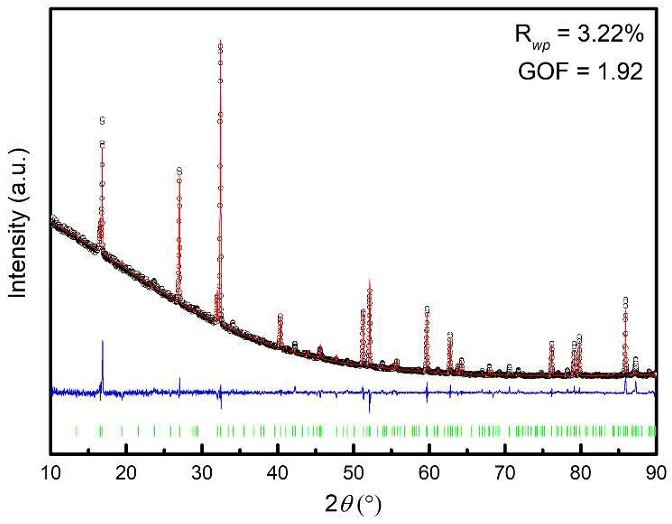 Figure 6. Rietveld refinement of laboratory X-ray diffraction data of Li4P2S6, showing the experimental data (black data points), the fit (red line), the difference profile (blue line) and expected Bragg reflections. The resulting profile residuals are given in the inset. (Neuberger et al., 2018)
Figure 6. Rietveld refinement of laboratory X-ray diffraction data of Li4P2S6, showing the experimental data (black data points), the fit (red line), the difference profile (blue line) and expected Bragg reflections. The resulting profile residuals are given in the inset. (Neuberger et al., 2018)
Hyperpolarization Techniques
Hyperpolarization techniques have revolutionized NMR spectroscopy by significantly enhancing signal intensity, thereby broadening the scope of NMR applications in fields such as materials science, pharmaceuticals, and biological research. In addition to Dynamic Nuclear Polarization (DNP), other methods such as Parahydrogen Induced Polarization (PHIP) and Signal Amplification by Reversible Exchange (SABRE) have emerged as powerful signal enhancement tools.
Parahydrogen-Induced Polarization (PHIP)
PHIP exploits the unique spin properties of parahydrogen, a specific nuclear spin isomer of hydrogen, to enhance NMR signals. The technique involves hydrogenating unsaturated substrates with parahydrogen, which transfers its spin order to the target molecules. This process can result in significant signal enhancement, enabling real-time monitoring of chemical reactions and metabolic processes.
Applications of PHIP
- Chemical Reaction Monitoring: PHIP allows for the detailed observation of reaction kinetics and mechanisms in real-time.
- Metabolic Studies: It can be used to track metabolic pathways in biological systems by enhancing the NMR signals of metabolites.
- Drug Discovery: PHIP can improve the sensitivity of NMR spectroscopy in studying small molecules and peptides, which is crucial for drug development.
Signal Amplification by Reversible Exchange (SABRE)
SABRE is another innovative hyperpolarization technique that transfers spin polarization from parahydrogen to target molecules without requiring chemical modification. This method is particularly advantageous for enhancing the NMR signals of biomolecules, making it a valuable tool for metabolic studies and molecular imaging.
Mechanism of SABRE
- SABRE relies on the reversible binding of parahydrogen to a metal catalyst, which then transfers its spin polarization to the target molecules.
- This process can be repeated multiple times, leading to significant signal amplification.
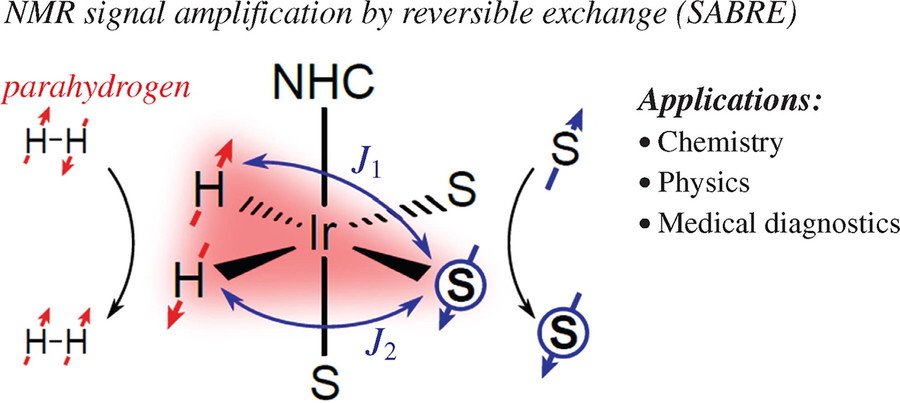 Figure 7. Parahydrogen-based NMR signal amplification by reversible exchange (SABRE) (Salnikov et al., 2023)
Figure 7. Parahydrogen-based NMR signal amplification by reversible exchange (SABRE) (Salnikov et al., 2023)
Applications of SABRE
- Biomolecular Imaging: SABRE can enhance the NMR signals of biomolecules, facilitating their detection and imaging in complex biological environments.
- Metabolic Studies: It can be used to study metabolic processes by enhancing the signals of low-concentration metabolites.
- Drug Development: SABRE can improve the sensitivity of NMR spectroscopy in studying drug candidates, thereby accelerating the drug discovery process.
Ultrafast 2D NMR Spectroscopy
Traditional 2D NMR methods require multiple scans to collect data in the indirect dimension, which can be time-consuming and impractical for dynamic samples or real-time monitoring. In contrast, UF 2D NMR can generate complete 2D spectra in a single scan, often within seconds
Principles and Advantages
The key innovation of UF 2D NMR is the replacement of the conventional time-increment approach with spatial encoding. In this method, spins located at different positions within the sample undergo different evolution periods simultaneously. This spatial encoding is achieved using gradient-based techniques that allow the acquisition of multidimensional correlation data in a single transient.
- Rapid Acquisition: UF 2D NMR can produce spectra in subseconds, making it suitable for real-time monitoring of chemical reactions and metabolic processes.
- Compatibility with Dynamic Systems: Its single-scan nature allows for the study of samples whose composition evolves over time, such as in reaction kinetics and chromatography.
- Enhanced Versatility: UF 2D NMR can be applied to both liquid and anisotropic media, including solid-state NMR studies.
- Integration with Other Techniques: It can be coupled with hyperpolarization methods and chromatography, further expanding its applications.
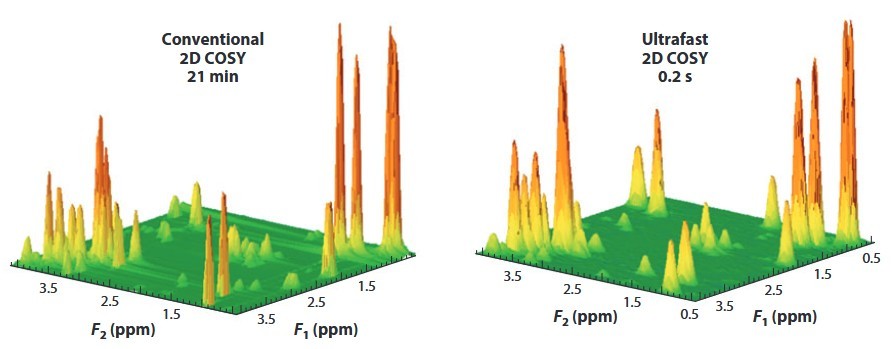 Figure 8. (a) Conventional and (b) UF 2D 1H homonuclear spectra (COSY) recorded on a mixture of metabolites on a 500-MHz Avance III spectrometer equipped with a cryoprobe. Abbreviations: COSY, correlation spectroscopy; UF, ultrafast. (Giraudeau and Frydman, 2014)
Figure 8. (a) Conventional and (b) UF 2D 1H homonuclear spectra (COSY) recorded on a mixture of metabolites on a 500-MHz Avance III spectrometer equipped with a cryoprobe. Abbreviations: COSY, correlation spectroscopy; UF, ultrafast. (Giraudeau and Frydman, 2014)
Applications in Real-Time Analysis
- Real-Time Reaction Monitoring: UF 2D NMR enables the detailed observation of reaction mechanisms and kinetics in organic and biochemistry.
- Metabolic Studies: It can track metabolic pathways in biological systems by enhancing the sensitivity of NMR spectroscopy.
- Quantitative NMR (qNMR): UF 2D NMR is particularly useful in "omics" disciplines, such as metabolomics, where rapid and quantitative analysis is crucial.
- Material Science: The technique can be applied to study the dynamics and structure of materials in real-time.
Advanced NMR Pulse Sequences
Innovations in NMR pulse sequence design have enhanced the capability to extract specific information from complex systems.
- Non-Uniform Sampling (NUS): NUS techniques involve the collection of a subset of data points in multidimensional NMR experiments, followed by computational reconstruction. This approach reduces acquisition time and improves sensitivity, allowing the study of larger biomolecules and faster processes.
- Relaxation Dispersion Experiments: Relaxation dispersion experiments measure conformational dynamics by monitoring changes in relaxation rates under varying conditions. These experiments provide insight into molecular motion on microsecond to millisecond timescales, which is critical for understanding enzyme mechanisms and protein folding.
Integration with Other Analytical Techniques
Combining NMR with complementary analytical methods has expanded its application and provided more comprehensive data.
- NMR-Mass Spectrometry (MS) Hyphenation: The integration of NMR with MS allows structural information to be correlated with molecular mass and composition. This combination is particularly valuable in metabolomics and complex mixture analysis, providing a more complete understanding of sample constituents.
- NMR-Microscopy: NMR microscopy combines magnetic resonance imaging (MRI) with high-resolution NMR spectroscopy to visualize molecular structures in intact samples. This technique allows the study of the spatial distribution of compounds in heterogeneous materials such as tissues and polymers.
Artificial Intelligence and Machine Learning in NMR
The integration of artificial intelligence (AI) and machine learning (ML) into NMR spectroscopy has revolutionized data analysis, providing greater accuracy and efficiency in interpreting complex data sets.
- Automated Spectral Assignment: ML algorithms have been developed to automate the process of spectral assignment, a traditionally labor-intensive task. By training models on large NMR datasets, these algorithms can accurately identify chemical shifts and coupling patterns, speeding structural elucidation.
- Predictive Modeling: AI-driven predictive models can predict NMR spectra based on molecular structures, helping to verify synthesized compounds and identify unknowns. This capability is particularly valuable in drug discovery and materials science, where rapid characterization is essential.
Select Service
At Creative Biostructure, we are committed to providing the most advanced NMR solutions to drive your research and development forward. With the latest innovations in NMR technology, we offer unparalleled capabilities for structural elucidation, metabolomics, drug discovery, and materials characterization. Our state-of-the-art instruments and expert support ensure that you get the most out of your NMR experiments. Contact us today to learn more and take the next step in your scientific journey.
References
- Giraudeau P, Frydman L. Ultrafast 2D NMR: an emerging tool in analytical spectroscopy. Annu Rev Anal Chem (Palo Alto Calif). 2014;7:129-161. doi:10.1146/annurev-anchem-071213-020208
- Lilly Thankamony AS, Wittmann JJ, Kaushik M, Corzilius B. Dynamic nuclear polarization for sensitivity enhancement in modern solid-state NMR. Progress in Nuclear Magnetic Resonance Spectroscopy. 2017;102-103:120-195. doi:10.1016/j.pnmrs.2017.06.002
- Martineau C. NMR crystallography: Applications to inorganic materials. Solid State Nuclear Magnetic Resonance. 2014;63-64:1-12. doi:10.1016/j.ssnmr.2014.07.001
- Neuberger S, Culver SP, Eckert H, Zeier WG, Schmedt auf der Günne J. Refinement of the crystal structure of Li4P2S6 using NMR crystallography. Dalton Trans. 2018;47(33):11691-11695. doi:10.1039/C8DT02619J
- Salnikov OG, Burueva DB, Skovpin IV, Koptyug IV. Parahydrogen-based NMR signal amplification by reversible exchange (Sabre): Recent advances and applications. Mendeleev Communications. 2023;33(5):583-596. doi:10.1016/j.mencom.2023.09.001
- Toke O. Three decades of REDOR in protein science: a solid-state NMR technique for distance measurement and spectral editing. IJMS. 2023;24(17):13637. doi:10.3390/ijms241713637