Natural products derived from plants, microorganisms, and marine organisms play an important role in drug discovery, agriculture, and biotechnology. Their complex and diverse structures, such as alkaloids, terpenes, and polyketides, pose significant challenges for identification. Accurate structure elucidation is essential for understanding bioactivity, optimizing synthesis, and ensuring safety. Nuclear magnetic resonance (NMR) spectroscopy is a cornerstone technique in this endeavor, providing unparalleled insight into molecular structures without destroying the sample.
Creative Biostructure offers professional NMR spectroscopy services, including solid-state NMR, solution-state NMR, quantitative NMR, and more, to help you identify natural products for research and industry.

Introduction of Natural Compounds
Natural compounds are chemical substances that occur naturally in nature, primarily derived from plants, animals, fungi, and microorganisms. These compounds have diverse biological activities and are essential in various fields, including medicine, agriculture, and industry.
Natural compounds can be broadly categorized into alkaloids, terpenes, flavonoids, phenolic compounds, glycosides, lipids, and proteins, among others. Many of these compounds have evolved as part of the organisms' defense mechanisms against predators, pathogens, or environmental stressors, and they often exhibit unique pharmacological properties.
- Alkaloids: These nitrogen-containing compounds often have powerful physiological effects on humans and animals. Examples include morphine (from the opium poppy) and quinine (from the cinchona tree).
- Terpenes: These compounds are produced by plants and have a wide range of uses, from fragrances and flavors to medicinal applications. Examples include menthol and camphor.
- Flavonoids: Found in fruits, vegetables, and herbs, flavonoids have antioxidant, anti-inflammatory, and anticancer properties. Common examples include quercetin and catechins.
- Phenolic Compounds: These include a wide range of substances like tannins and lignans, known for their antioxidant activities and potential health benefits.
- Glycosides: These compounds are sugars bound to other functional groups and are often used in medicine. For example, cardiac glycosides like digoxin are used to treat heart conditions.
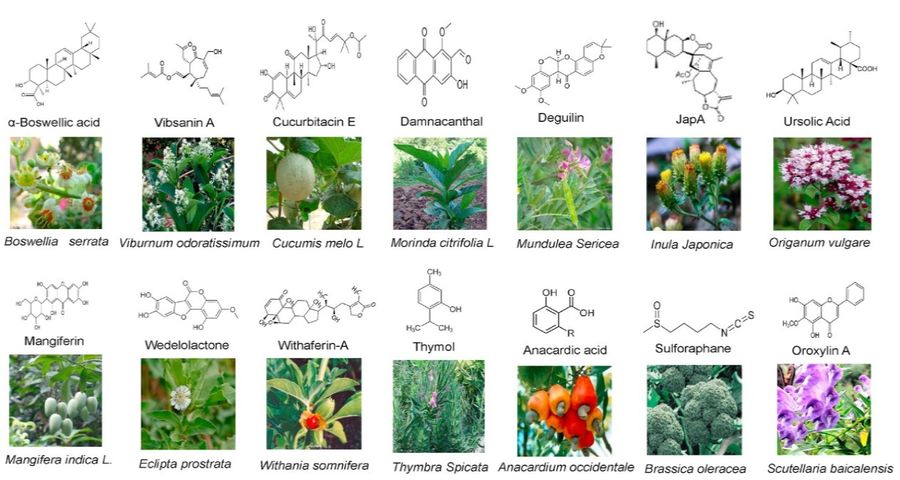 Figure 1. Structures of various natural compounds and images of their source medicinal plants. (Tsolmongyn et al., 2013)
Figure 1. Structures of various natural compounds and images of their source medicinal plants. (Tsolmongyn et al., 2013)
Techniques to Identifying and Analyzing Natural Compound
| Chromatography | Chromatography techniques separate compounds based on their interaction with a stationary phase and a mobile phase. They are often the first step in identifying natural compounds. |
| Mass Spectrometry (MS) | Mass spectrometry is a powerful technique for determining the molecular weight and structure of compounds. |
| Nuclear Magnetic Resonance (NMR) Spectroscopy | NMR is used to determine the structure of organic compounds by analyzing the interaction of nuclear spins with magnetic fields. |
| Infrared Spectroscopy (IR) | IR spectroscopy is used to identify functional groups within a compound. The sample absorbs infrared radiation, and the resulting spectrum indicates the presence of bonds like C-H, N-H, C=O, and others based on their vibrational frequencies. |
| Ultraviolet-Visible (UV-Vis) Spectroscopy | UV-Vis spectroscopy is used to identify compounds that absorb UV or visible light, typically providing information about the conjugation of double bonds in organic compounds. It's particularly useful for the analysis of flavonoids, phenols, and alkaloids. |
| X-ray Crystallography | X-ray crystallography is a powerful technique for determining the 3D structure of crystalline compounds. It is particularly useful for large molecules such as proteins, complex natural products, or secondary metabolites. |
| Electrochemical Analysis | Electrochemical methods, such as cyclic voltammetry, can be used to study the redox behavior of natural compounds, especially those with antioxidant or electron transfer properties. |
| NMR Imaging and Relaxation Time Studies | This method is particularly useful for imaging large biomolecules in solution or in vivo. It can help determine the conformation and dynamic behavior of complex natural compounds. |
| Fluorescence Spectroscopy | Fluorescence is used to identify compounds that emit light when excited. This technique is useful for detecting compounds such as flavonoids, alkaloids, and certain enzymes that naturally fluoresce. |
| Bioassays | In addition to physical and chemical techniques, biological assays are often used to identify natural compounds with potential pharmacological activity. For example, antimicrobial, antioxidant, anti-inflammatory, or anticancer activities can be tested using cell cultures or animal models. This helps in identifying compounds with bioactive properties for drug development. |
Basics of NMR Spectroscopy
Principles of NMR
NMR exploits the magnetic properties of atomic nuclei (e.g., 1H, 13C) in a strong magnetic field. When exposed to radiofrequency pulses, the nuclei resonate at frequencies that depend on their chemical environment. The resulting spectrum provides a "fingerprint" of the molecule.
Chemical Shifts
Chemical shifts (measured in ppm) indicate the electronic environment of nuclei. For example:
- 1H NMR: Aromatic protons (6–8 ppm), aldehydes (9–10 ppm).
- 13C NMR: Carbonyl carbons (170–220 ppm), aliphatic carbons (0–100 ppm).
Spin-Spin Coupling
Splitting patterns in 1H NMR reveal adjacent protons. Coupling constants (J values) help determine stereochemistry (e.g., trans vs. cis alkenes).
NMR Techniques in Structure Elucidation
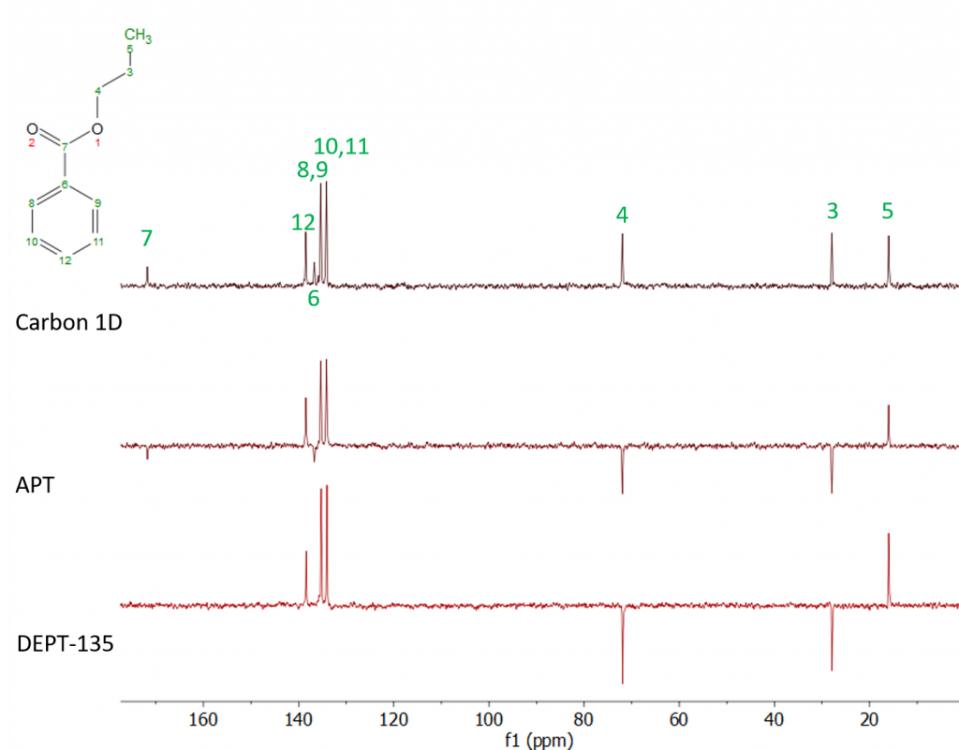
| 1D NMR Techniques | 1H NMR (Proton NMR) | 1H NMR is one of the most widely used NMR techniques for structural elucidation. It provides crucial information about the types of hydrogen (proton) environments in a molecule. Chemical shifts indicate the electronic environment of the protons, while integration reveals the relative number of each type of proton. Splitting patterns (multiplicity) arise from spin-spin coupling with neighboring protons, helping to establish connectivity and molecular structure. |
| 13C NMR (Carbon-13 NMR) | 13C NMR is essential for determining the carbon skeleton of a molecule. Unlike 1H NMR, 13C NMR spectra are typically simpler due to the low natural abundance of 13C and the lack of strong spin-spin coupling. Chemical shifts provide insight into the types of carbon environments present (e.g. aliphatic, aromatic, carbonyl, etc.). Techniques such as broadband decoupling can be used to remove splitting from directly attached protons, resulting in clearer spectra. | |
| DEPT (Distortionless Enhancement by Polarization Transfer) | DEPT experiments are used to distinguish different types of carbon atoms (CH3, CH2, CH, and quaternary carbons). This technique enhances the sensitivity of 13C NMR and provides additional structural information. | |
| 2D NMR Techniques | COSY (Correlation Spectroscopy) | COSY is a homonuclear two-dimensional experiment that maps the scalar coupling between protons. It provides information about protons that are spin-spin coupled, usually those that are adjacent in a molecule. Cross peaks in a COSY spectrum indicate which protons are coupled, helping to establish connectivity and fragment structures. |
| HSQC (Heteronuclear Single Quantum Coherence) | HSQC is a heteronuclear 2D experiment that provides direct correlations between 1H and 13C nuclei. Each cross-peak in an HSQC spectrum corresponds to a 1H nucleus directly bonded to a 13C nucleus, making it an efficient tool for assigning carbon chemical shifts and aiding in structural determination. | |
| HMBC (Heteronuclear Multiple Bond Correlation) | HMBC detects long-range (two- to four-bond) correlations between protons and carbon atoms. Unlike HSQC, which only shows direct 1H-13C connectivity, HMBC allows for the identification of more distant relationships in the molecular framework. This technique is particularly useful for identifying functional groups, establishing connectivity across ring systems, and deducing complex structures. | |
| NOESY (Nuclear Overhauser Effect Spectroscopy) | NOESY provides spatial information about molecular structures by detecting dipolar interactions between protons that are within 5 Å of each other. This technique is especially valuable for determining the three-dimensional conformation of molecules and stereochemistry. It helps identify through-space correlations rather than through-bond interactions, making it useful for studying macromolecules, cyclic compounds, and chiral centers. |
Application of NMR in Identifying Natural Compounds
Determining Molecular Framework
NMR is a powerful tool for identifying the core structures of natural compounds by analyzing chemical shifts, coupling patterns, and multiplicity. For example, terpene skeletons can be confirmed based on their characteristic proton and carbon resonances. In steroid structures, methyl groups often appear as singlets in 1H NMR, aiding in the identification of these complex frameworks.
Elucidating Stereochemistry
- Coupling Constants: The magnitude of the J coupling constants helps distinguish axial and equatorial protons in cyclic systems such as cyclohexanes. Axial protons have larger coupling constants (J = 10-14 Hz), while equatorial protons have smaller values (J = 2-4 Hz), providing important insight into stereochemical arrangements.
- NOE (Nuclear Overhauser Effect) Effects: NOE experiments detect spatial proximity between protons, allowing confirmation of cis/trans isomers and overall molecular configuration. This is particularly useful for complex natural product structures where stereochemistry is critical to bioactivity.
Identifying Functional Groups
- Hydroxyl Groups: Appear in the range of 1–5 ppm in 1H NMR, often showing broad signals due to exchangeable protons.
- Carbonyls Groups: Typically resonate between 200–220 ppm in 13C NMR, distinguishing ketones, aldehydes, and carboxyl groups.
- Aromatic and Aliphatic Functionalities: Proton and carbon signals provide further characterization, distinguishing between sp2 and sp3 hybridized carbons.
Quantitative Analysis (qNMR)
Quantitative NMR (qNMR) is an essential technique for determining the purity and concentration of natural compounds. By integrating peak areas, qNMR enables:
- Purity Assessment: Used in the pharmaceutical and nutraceutical industries to ensure batch-to-batch consistency.
- Mixture Analysis: Determines the proportions of various components in complex mixtures, useful for herbal extracts and fermentation products.
- Standardization: Provides reliable quantification for bioactive natural products, supporting regulatory requirements and quality control.
Complementary Techniques and Hyphenated Methods
LC-NMR
Liquid chromatography (LC) is an essential separation technique in natural product research, effectively resolving complex mixtures of bioactive compounds. When coupled with NMR, LC-NMR allows direct structural analysis without the need for time-consuming isolation and purification steps. This is particularly useful when studying crude extracts from plants, fungi or marine organisms, where several structurally related compounds may coexist.
LC-NMR operates in either off-line, on-flow, or stop-flow modes. Off-line LC-NMR collects fractions after chromatographic separation for subsequent NMR analysis. On-flow LC-NMR records spectra as separated components elute through the NMR detector, providing rapid insight but requiring high sensitivity. Stop-flow LC-NMR stops the flow at specific times to capture detailed spectra, balancing resolution and throughput.
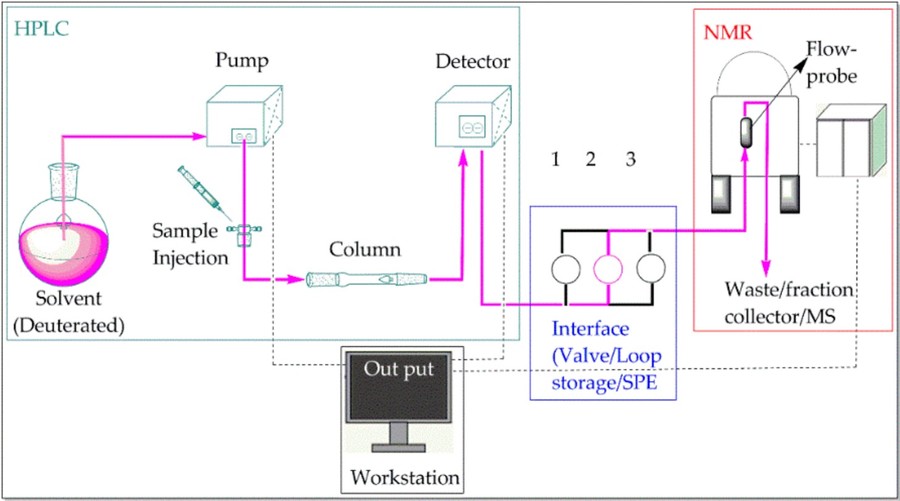 Figure 2. The instrumental setup of LC–NMR system. (Gebretsadik, 2021)
Figure 2. The instrumental setup of LC–NMR system. (Gebretsadik, 2021)
The advantages of LC-NMR include reduced sample preparation, the ability to analyze unstable or low-yield compounds, and minimized sample loss. However, challenges such as solvent suppression, limited sensitivity, and the need for specialized instrumentation must be addressed to maximize its efficacy.
MS-NMR Integration
Mass spectrometry (MS) is another indispensable tool for natural product identification, excelling at determining molecular weight, elemental composition, and fragmentation patterns. However, MS alone often falls short in fully elucidating structural motifs, particularly stereochemistry and connectivity. Integrating MS with NMR overcomes these limitations by providing both molecular weight information and detailed structural insights.
In MS-NMR workflows, mass spectrometry is typically used first to screen complex mixtures, identifying target compounds based on mass-to-charge ratios and isotopic patterns. Once a compound of interest is detected, NMR analysis is performed to determine connectivity, functional groups, and three-dimensional conformation. This synergy accelerates the dereplication process-rapidly distinguishing novel compounds from known ones in natural product libraries-thereby streamlining drug discovery and metabolomics research.
Recent advances in hyphenated MS-NMR techniques, such as direct infusion MS-NMR and solid-phase microextraction (SPME) MS-NMR, further enhance sensitivity and sample throughput. Despite technical challenges such as sample concentration limitations and solvent compatibility, MS-NMR integration remains an invaluable approach for comprehensive compound characterization.
Case Study
Case 1: Extraction, Purification, and NMR Analysis of Terpenes from Brown Algae
Algal terpenes are a diverse group of marine natural products that differ from their terrestrial counterparts. Brown algae, especially those of the class Phaeophyceae, are rich sources of biologically significant terpenoids, mainly diterpenes and meroditerpenes. This study outlines the analytical methods for the isolation and characterization of these compounds from brown seaweeds. The process includes (1) extraction of the lipid content, (2) purification of the terpenes using chromatographic techniques, and (3) elucidation of their structures using 1D and 2D NMR.
 Figure 3. Expansion (olefinic protons region) of the 1H NMR spectrum (CDCl3, 500 MHz) of degraded methoxybifurcarenone (MBFC). (Stengel and Connan, 2015)
Figure 3. Expansion (olefinic protons region) of the 1H NMR spectrum (CDCl3, 500 MHz) of degraded methoxybifurcarenone (MBFC). (Stengel and Connan, 2015)
Case 2: Complementary Analytical Platforms of NMR Spectroscopy and LCMS Analysis in the Metabolite Profiling of Lsochrysis galbana
This study profiles the metabolites of Isochrysis galbana, a lesser-known microalga, using 1H NMR spectroscopy and LC-MS. Five different extracts (Hex, EtOAc, EtOH, AqE, and Aq) were analyzed, and PLS-DA revealed that EtOAc and Aq extracts contained the most diverse metabolites. A total of 21 metabolites, including carotenoids, polyunsaturated fatty acids, and amino acids, were identified from the NMR data, while LC-MS/MS annotated 32 metabolites, 10 of which overlapped with the NMR profile. In addition, 11 glycerophospholipids were discovered by MS/MS molecular networking. These results improve the understanding of the chemical composition of I. galbana and highlight its potential as an aquaculture feed ingredient.
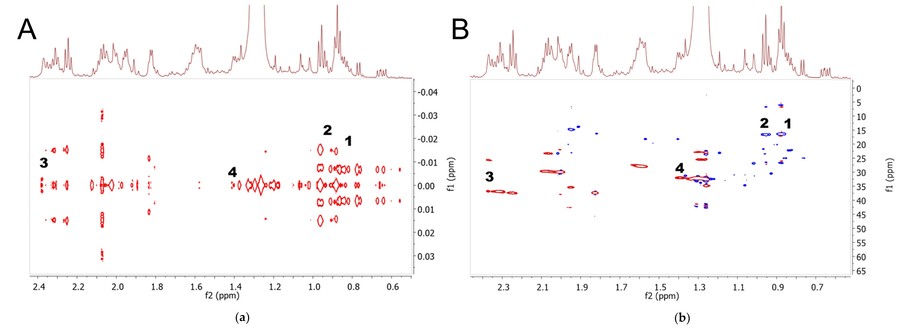 Figure 4. Representative 2D-J-resolved (A) and HSQC spectra (B) of EtOAc extract. The signals were assigned as follows: 1, non-PUFA; 2, PUFA; 3, DHA; and 4, DPA. (Bustamam et al., 2021)
Figure 4. Representative 2D-J-resolved (A) and HSQC spectra (B) of EtOAc extract. The signals were assigned as follows: 1, non-PUFA; 2, PUFA; 3, DHA; and 4, DPA. (Bustamam et al., 2021)
Select Service
Unlock the full potential of your natural compound research with our advanced NMR services. From structural elucidation to purity assessment, Creative Biostructure provides high-resolution 1H and 13C NMR, 2D NMR techniques, and quantitative analysis tailored to your needs. Partner with us for accurate, reliable, and efficient NMR solutions—contact us today to enhance your research!
References
- Bustamam MSA, Pantami HA, Azizan A, et al. Complementary analytical platforms of NMR spectroscopy and LCMS analysis in the metabolite profiling of Isochrysis galbana. Marine Drugs. 2021;19(3):139. doi:10.3390/md19030139
- Gebretsadik T, Linert W, Thomas M, Berhanu T, Frew R. LC–NMR for natural product analysis: a journey from an academic curiosity to a robust analytical tool. Sci. 2021;3(1):6. doi:10.3390/sci3010006
- Stengel DB, Connan S. Marine algae: a source of biomass for biotechnological applications. In: Stengel DB, Connan S, eds. Natural Products from Marine Algae. Vol 1308. Springer New York; 2015:1-37. doi:10.1007/978-1-4939-2684-8_1
- Tsolmongyn B, Koide N, Odkhuu E, et al. Lipopolysaccharide prevents valproic acid-induced apoptosis via activation of nuclear factor-κB and inhibition of p53 activation. Cellular Immunology. 2013;282(2):100-105. doi:10.1016/j.cellimm.2013.04.011